 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Progress Toward Poliomyelitis Eradication -- Eastern Mediterranean Region, 1988-1994In 1988, the Regional Committee of the Eastern Mediterranean Region (EMR) of the World Health Organization * (WHO) adopted a resolution to eradicate poliomyelitis from the region by the year 2000 (1). Since this goal was established, substantial progress toward polio eradication has been achieved using three major strategies: 1) achieving and maintaining high coverage with at least three doses of oral poliovirus vaccine (OPV3); 2) implementing supplementary vaccination activities, including National Immunization Days (NIDs) **, to rapidly interrupt poliovirus transmission; and 3) developing sensitive systems of epidemiologic and laboratory surveillance, including use of the standard WHO case definition (2). *** This report summarizes progress toward polio eradication in EMR countries from 1988 through 1994 and is based on reports received through August 1, 1995. Incidence of Polio From 1988 through 1994, the number of confirmed polio cases reported in the region decreased 57%, from 2342 to 1015 Figure_1. The sharp decline in reported cases from 1993 (2451 cases) to 1994 (1015 cases) especially reflected improved control of polio in Pakistan and Sudan, both of which experienced large outbreaks in 1993. Both countries conducted NIDs for the first time in 1994. Despite the substantial decrease in the number of cases reported from 1993 to 1994, Pakistan continues to report more cases than any other country in the region; the 527 cases reported in 1994 represent 52% of the regional total. During 1994, nine other countries reported polio cases, including Yemen (173 {17%}), Egypt (120 {10%}), Iran (93 {9%}), Iraq (63 {6%}), Sudan (25 {2%}), Saudi Arabia (six {0.5%}), Jordan (four {0.3%}), and Lebanon and Syria (one each {0.1%}). Eleven countries reported no cases. Five countries (Cyprus, Kuwait, Libya, Morocco, and Qatar) have reported no cases for at least 3 years. Vaccination Coverage From 1990 through 1993, routine coverage with OPV3 among children aged less than 1 year in EMR was greater than or equal to 80%; coverage decreased in 1994 to 78% Figure_1. Of 20 countries reporting OPV3 coverage in 1994, a total of 16 reported coverage greater than 80%. Of these, 12 reported OPV3 coverage greater than 90%. The decrease in coverage in 1994 primarily reflected declining coverage in four countries (Djibouti, Pakistan, Sudan, and Yemen). In conjunction with declining routine vaccination coverage, Pakistan experienced an outbreak of paralytic polio in the second and third quarters of 1995 in its most populous province (Punjab). NIDs were conducted in two countries (Egypt and Syria) in 1993 and in five countries (Egypt, Iran, Pakistan, Sudan, and Syria) in 1994. By the end of 1995, a total of 19 (82%) countries, representing 93% of the estimated population in the region, will have conducted NIDs Figure_2. Cyprus, Djibouti, Somalia, and Yemen will not conduct NIDs in 1995. Surveillance By 1992, a total of 21 of the 23 countries in EMR had developed surveillance for polio; surveillance could not be established in Afghanistan and Somalia because of civil conflict. By 1994, a total of 19 countries had established systems for reporting and monitoring the occurrence of acute flaccid paralysis (AFP), compared with six countries in 1992. Four countries (Afghanistan, Pakistan, Somalia, and Yemen) are not reporting cases of AFP. By 1994, AFP or polio cases were being investigated clinically and epidemiologically in 21 countries, compared with eight in 1992. An important indicator of the sensitivity of surveillance that has been consistent in many countries throughout the world is the rate of nonpolio AFP among children aged less than 15 years (greater than or equal to 1 case per 100,000); by 1994, five EMR countries had achieved this rate. Laboratory Support By 1994, laboratory surveillance for polioviruses had been initiated in 14 countries, compared with six in 1992. In 1994, the EMR laboratory network was involved in the investigation of 717 AFP cases. Of these, two stool specimens were available for 495 (69%). Of 422 cases with onset and collection data available, 354 (84%) had stool specimens collected within 14 days of onset of paralysis. Reported by: Regional Office for the Eastern Mediterranean Region, Alexandria, Egypt; Global Program for Vaccines and Immunization, World Health Organization, Geneva. Respiratory and Enterovirus Br, National Center for Infectious Diseases; Polio Eradication Activity, National Immunization Program, CDC. Editorial NoteEditorial Note: The findings in this report document substantial progress toward polio eradication in EMR during 1988-1994. In addition to providing member countries with technical support to implement global polio eradication strategies, the EMR has initiated three major regional initiatives to accelerate polio eradication. First, during 1993-1994, WHO collaborated with Rotary International and the United Nations Children's Fund (UNICEF) and used teams of national and international laboratory and epidemiology experts to conduct rapid surveillance assessments in 19 countries. Second, WHO coordinated efforts (including NIDs) to detect and prevent cases in three geographically contiguous and epidemiologically similar countries (the Mahgreb Union in Northern Africa; the Arab Gulf states; and the Middle Eastern and Asian states of Jordan, Lebanon, Palestine, Syria, Afghanistan, Iran, Iraq, and Pakistan) to foster the emergence of polio-free zones within the region. Third, a monthly newsletter ("Poliofax") is sent to all ministries of health in the region to provide feedback and to encourage complete and timely disease reporting. Despite progress toward polio eradication, some barriers persist and underscore the need to 1) increase vaccination levels in unvaccinated subpopulations; 2) maintain high routine vaccination coverage in all member countries; 3) translate political commitment into action, including the provision of sufficient funds for the purchase of oral polio vaccine (both for routine and supplementary vaccination) by governments of all member countries; 4) encourage all member countries to establish polio eradication as a priority activity, including the initiation of AFP surveillance and implementation of NIDs; 5) overcome the impediments to the regional polio eradication initiative as the result of war and civil strife in some countries; and 6) strengthen the timely exchange of information among countries and with WHO, UNICEF, Rotary International, and other partner organizations to enable coordination and enhanced support of regional polio eradication activities. References
* Member countries are Djibouti, Egypt, Libya, Morocco, Somalia, Sudan, and Tunisia in northern and eastern Africa; the Arab Gulf states of Bahrain, Kuwait, Oman, Qatar, Saudi Arabia, United Arab Emirates, and Yemen; Iraq, Jordan, Lebanon, Syria, and the Palestinian people in the Middle East; Afghanistan, Iran, and Pakistan in Asia; and Cyprus. ** Mass campaigns over a short period (days to weeks) in which two doses of OPV are administered to all children in the target age group (usually age less than 5 years) regardless of prior vaccination history, with an interval of 4-6 weeks between doses. *** A confirmed case of polio is defined as acute flaccid paralysis and at least one of the following: 1) laboratory-confirmed wild poliovirus infection, 2) residual paralysis at 60 days, 3) death, or 4) no follow-up investigation at 60 days. Figure_1 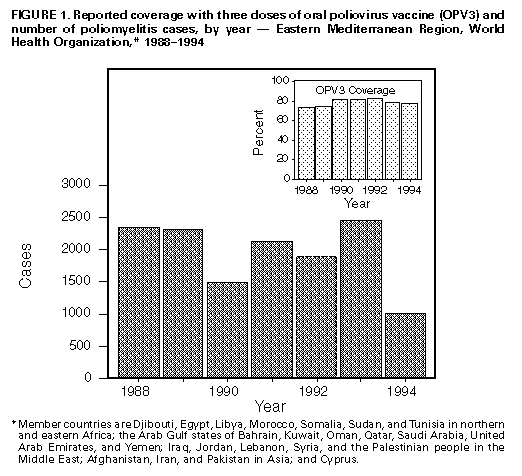 Return to top. Figure_2 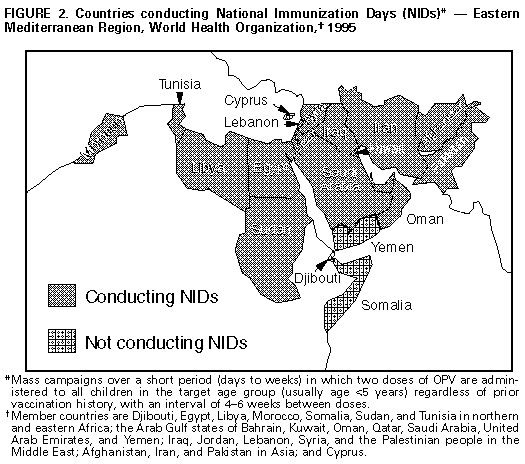 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 09/19/98 |
|||||||||
This page last reviewed 5/2/01
|