 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Current Trends Impact of Missed Opportunities to Vaccinate Preschool-Aged Children on Vaccination Coverage Levels -- Selected U.S. Sites, 1991-1992Vaccination coverage levels among 2-year-old children for each of three routinely recommended vaccines -- diphtheria and tetanus toxoids and pertussis (DTP), oral poliovirus (OPV), and measles-mumps-rubella (MMR) -- are lower than the national Childhood Immunization Initiative (CII) goal of 90% coverage for these vaccines (1,2). During 1991-1992, CDC awarded contracts to four universities (in Philadelphia, Los Angeles, Baltimore, and Rochester, New York) to conduct evaluations to identify causes of undervaccination, characterize and quantify missed opportunities (MOs) to vaccinate, and assess their programmatic importance. The evaluations targeted high-risk racial/ethnic minority children in inner-city settings in the four urban sites. This report summarizes selected findings * from these studies. For each study, the proportion of health-care visits with an MO were determined through assessments of clinic medical records. An MO was defined as a health-care visit during which a child eligible for vaccination on the day of the visit and with no contraindication for vaccination failed to receive the needed dose(s). By assuming that all types of MOs (e.g., not assessing the vaccination status of children during visits, not administering needed vaccines because of the presence of a medical condition inaccurately perceived as a contraindication, and not administering needed vaccines simultaneously) had been eliminated, hypothetical coverage levels were calculated at ages 12 and 24 months for individual vaccines. Based on medical records, at least one MO occurred for 377 (75%) of 502 children in Baltimore, 518 (69%) of 752 in Los Angeles, 621 (64%) of 971 in Philadelphia, and 440 (82%) of 534 in Rochester. Of the total 25,139 health-care visits evaluated, 5163 (21%) were associated with at least one MO. MOs occurred during both sick- and well-child care visits but were more likely to occur during sick-child visits. For example, in Rochester, 23% of all MOs for the receipt of a fourth dose of DTP/diphtheria and tetanus toxoids (DTP/DT) occurred during well-child visits, 22% occurred during follow-up visits, and 55% occurred during sick-child visits. In Baltimore, MOs were nearly three times as likely to occur during sick-child visits (even in the absence of contraindications) (85%) than during well-child care visits (30%). Failure to administer all indicated vaccines simultaneously (e.g., administering DTP/DT, poliovirus vaccine, and MMR together when indicated) on the day of the visit accounted for 12% of all MOs in Baltimore, 9% in Los Angeles, and 3% in Rochester. In Philadelphia, failure to administer vaccines simultaneously accounted for 1% (for two doses of DTP/DT) to 15% (for four doses of DTP/DT) of all MOs. Hypothetical coverage levels for individual vaccines were calculated at ages 12 and 24 months for all surveyed children. Based on these calculations, coverage levels at age 12 months for three doses of DTP/DT and two doses of poliovirus vaccine would have increased by 4-27 percentage points in all four sites (Table_1). Coverage levels at age 24 months varied by site and vaccine; for three doses of DTP/DT and MMR, coverage levels would have increased by less than 10 percentage points in all sites. In comparison, for four doses of DTP/DT, coverage would have increased by 16 percentage points in Baltimore (from 58% to 74%), eight percentage points in Los Angeles (from 26% to 34%), 12 percentage points in Philadelphia (from 57% to 69%), and 21 percentage points in Rochester (from 75% to 96%). For three doses of poliovirus vaccine, increases in coverage would have ranged from five percentage points in Rochester to 16 percentage points in Baltimore and Los Angeles. In the sites that also calculated hypothetical coverage levels among surveyed children who were not up-to-date at age 24 months with the 4:3:1 combined series ** (Baltimore, Los Angeles, and Rochester), coverage levels at age 24 months would have increased (Figure_1). Among these children, elimination of MOs resulted in greater absolute increases in coverage levels (range: 12-80 percentage points; median: 20 percentage points), compared with elimination of MOs among all surveyed children (range: 3-21 percentage points; median: 10 percentage points). Reported by: S Zimicki, MA, S McCombie, PhD, C Koepke, MA, D Romer, PhD, R Hornik, PhD, A Arbeter, MD, Albert Einstein Medical Center, Philadelphia. D Wood, MD, M Pereyra, MPH, N Halfon, MD, JS Hamlin, MPH, Cedars-Sinai Medical Center, Los Angeles. E Holt, DrPh, D Guo, MS, N Hughart, MPH, V Keane, MD, B Stanton, MD, B Guyer, MD, The Johns Hopkins Univ, Baltimore. L Rodewald, MD, P Szilagyi, MD, S Humiston, MD, K Roghmann, PhD, R Raubertas, PhD, Univ of Rochester, Rochester, New York. National Immunization Program, CDC. Editorial NoteEditorial Note: The objectives of CII for 1996 are to increase vaccination coverage levels among 2-year-old children to at least 90% for one dose of MMR and three doses each of DTP, OPV, and Haemophilus influenza type b vaccine and to at least 70% for three doses or more of hepatitis B vaccine. In 1993, preliminary national coverage levels for these vaccines ranged from 55% to 88%, and levels generally were lower among children in inner-city settings (3). The findings of the four assessments in this report suggest that coverage could improve substantially by changing provider vaccination practices that result in MOs. During 1991-1992, at least one MO occurred for approximately half of all children surveyed in the four sites, highlighting the potential for improvement in coverage levels if all MOs had been eliminated. In particular, substantially greater improvements in coverage would have resulted from elimination of MOs among children who were not up-to-date (i.e., the group in greatest need of interventions). The variations in vaccination coverage by site may have reflected differences in health-care use patterns (i.e., the number of health-care contacts of a child). In addition, the impact on coverage levels of eliminating MOs may be dependent in part on existing coverage levels: as coverage increases, elimination of MOs may be associated with smaller increases in coverage. Other studies also have documented the impact of MOs (4-6). However, because the studies in this report primarily targeted high-risk racial/ethnic minority groups in inner-city settings, these findings may not be generalizable to all areas of the United States. To meet the objectives of CII for 1996, public and private health-care providers need to aggressively implement changes in their vaccination practices (e.g., those outlined in the Standards for Pediatric Immunization Practices {7}). In particular, changes to eliminate MOs to vaccinate include 1) maintaining accurate vaccination records, 2) assessing the vaccination status of children at every contact with the health-care system, 3) using only true medical contraindications (e.g., vaccination should not be deferred because of minor illness), and 4) administering needed vaccines simultaneously. References
* These studies were completed in mid-1993. Subsequent analysis of data about MOs was completed during late 1993. Analysis of data from these studies is ongoing. ** Four doses of DTP/DT, three doses of poliovirus vaccine, and one dose of MMR.
TABLE 1. Percentage of actual and hypothetical * vaccination coverage among all
surveyed children for individual vaccine doses, by age and site -- selected U.S. sites,
1991-1992
================================================================================================
Baltimore Los Angeles Philadelphia Rochester, N.Y.
---------------- ---------------- ---------------- ---------------
Age Vaccine/ Hypo- Hypo- Hypo- Hypo-
(mos) Dose Actual thetical Actual thetical Actual thetical Actual thetical
12 DTP/DT + /3 72 84 48 57 54 58 61 88
Polio & /2 86 92 64 70 71 77 88 96
24 DTP/DT/3 85 93 54 62 82 85 94 99
DTP/DT/4 58 74 26 34 57 67 75 96
Polio/3 65 81 34 50 68 79 80 95
MMR @ /1 80 89 39 48 87 94 90 96
------------------------------------------------------------------------------------------------
* Assumes all missed opportunities to vaccinate had been eliminated.
+ Diphtheria and tetanus toxoids and pertussis vaccine/diphtheria and tetanus toxoids.
& Poliovirus vaccine.
@ Measles-mumps-rubella vaccine.
=================================================================================================
Return to top. Figure_1 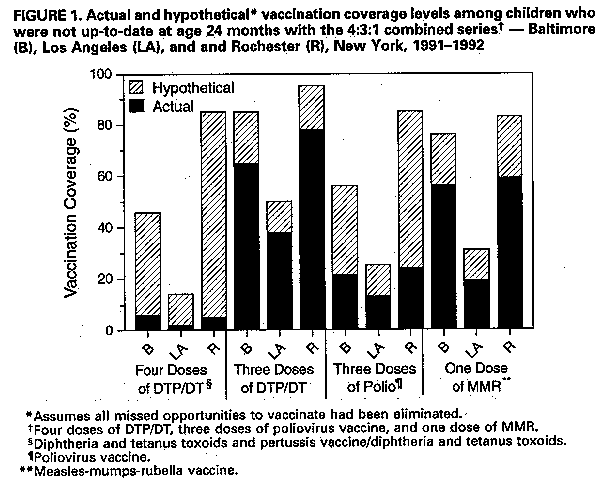 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 09/19/98 |
|||||||||
This page last reviewed 5/2/01
|