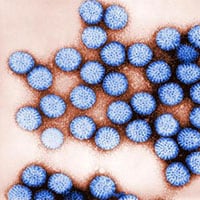
Collaborative Efforts Yield Improved Rotavirus Vaccine
Rotavirus is the most common diarrhea-causing pathogen in children. Prior to rotavirus vaccination availability, rotavirus caused an average of 611,000 deaths per year globally, and millions of hospitalizations. Virtually all children were infected by rotavirus by age 5. Rotavirus continues to cause severe diarrhea in infants and young children in the U.S. and globally. The majority of children that have access to adequate medical care survive infection with no significant long-term consequences. However, the number of deaths associated with severe diarrhea, vomiting, dehydration, and shock is staggering and preventive intervention is required. While rotavirus vaccines have proven effective in developed countries, the live attenuated oral approaches have been found to be significantly less effective in many resource-limited, developing countries where a vaccine is needed most.
CDC investigators have developed a method to inactivate rotavirus, while maintaining the structural integrity. This method provides a vaccine alternative to the live oral rotavirus vaccines. CDC partnered with Sanofi Pasteur through a Collaborative Research and Development Agreement (CRADA) and developed a new and improved heat inactivation method, instead of the more common formalin inactivation methods. The method developed by CDC is rapid, simple, and maintains the integrity of the virus particles and preserves their antigenicity. CDC holds a US patent on the technology, and has entered into licensing agreements with industry partners in China, India, and Vietnam. CDC has also entered into evaluation licenses with three US partners to evaluate CDC’s technology as part of their potential vaccine pipeline.