NSSP Update – September 2019

- Community of Practice
- NSSP Community of Practice Call
- Implementation Guide for Syndromic Surveillance
- Fall 2019 Pilot Testing of Implementation Guide
- CDC Funding Recipients and Partnerships
- NSSP Closes Out CoAg
- ESSENCE Queries

- Collaborations Accelerate SyS Practice
- Free-text Coding in NSSP-ESSENCE
- Data Quality Corner
- Introduction to NSSP–ESSENCE Data Quality Filters
- Spotlight on SyS Practice
People
Community of Practice
Monthly NSSP CoP Call

Please join your colleagues for the monthly NSSP Community of Practice (NSSP CoP) call. The next call will be held September 17, 2019, 3:00–4:30 PM ET. The topic is Earthquake Surveillance: Shaking Things Up with SyS.
New member? Syndrome? Success? Email NSSP!
Be part of the NSSP CoP. To participate, email nssp@cdc.gov and provide your name, contact information, and request to be part of the NSSP CoP. We’ll add your name to the membership list and send you invites to community activities.
If you have a syndrome to share, success story to showcase, or resources to post on the Knowledge Repository, please email nssp@cdc.gov. We will forward your request to the appropriate NSSP CoP committee or workgroup to review before posting.
Implementation Guide for Syndromic Surveillance
Final edits to the Implementation Guide for Syndromic Surveillance Release 1.0 were completed in June 2019 ahead of schedule, thanks to participation of Community of Practice members. In July 2019, the guide was posted on the HL7 website, and the National Institute of Standards and Technology Implementation Authoring Tool (NIST IGAMT) released the message validation tool for testing. NIST IGAMT is open access, and anyone can enter the site as a guest user and validate messages for this specification.
The ISDS and CDC Workgroup completed its revision of the Implementation Guide. The HL7 Public Health Workgroup approved the guide, launching a 90-day period for review so that all HL7 members have sufficient time to comment. The Implementation Guide will be posted as a WIKI page for collaborative editing. If you aren’t an HL7 member, you can make suggestions by submitting a ticket to the NSSP Service Desk (an account is required). NSSP has set up an Implementation Guide queue to triage comments. The WIKI is located at http://www.hl7.org/implement/standards/product_brief.cfm?product_id=503 and will be available to the general public for download on October 26, 2019. The 90-day comment period closes November 1, 2019.
| HL7 2.5.1 Implementation Guide Milestones* | |
|---|---|
| Time Frame | Activity |
| 2015 | Completed Version 2.0 Final RELEASE** |
| 2016 | Released Erratum and Clarification Documents for Version 2.0 |
| 2017 Summer | Released Version 2.2 Working Draft for Community Comment and Consensus |
| 2017 Winter | Released Version 2.3 for Review and Community Comment |
| 2018 March | Released Version .09 |
| 2018 Spring | Submitted DRAFT HL7 Guide for Balloting: Implementation Guide for Syndromic Surveillance Release 1.0 Standard for Trial Use (STU) HL7 Version 2.5.1 |
| 2018 Fall (October–December) |
|
| 2018 November– 2019 March |
|
| 2019 January | Provided Final Resolution for 33 Comments (Pending) |
| 2019 February |
|
| 2019 March–May |
|
| 2019 June 20–27 | Hand-off to HL7 Public Health Workgroup for 2-week Review |
| 2019 July |
|
| 2019 August–October |
|
| October 26, 2019 | Release to Public: HL7 2.5.1 Implementation Guide for Syndromic Surveillance Version 1 as a Standard for Trial Use for the General Public |
| 2019 September–November |
|
| 2019 September–2022 September | Publish Standard for Trial Use for Period of 1 to 3 years (during which time additional comments may be submitted and dispositioned and additional releases of the guide published) |
* Milestones revised September 4, 2019.
** Version 2.0 is currently being used; subsequent versions are working drafts only.
Shaded activities have been completed.
Fall 2019 Pilot Testing of Implementation Guide

Let’s kick the tires and take the Implementation Guide for a spin. In the next few months, NSSP will lead pilot tests for EHR vendors, hospitals, and health departments. About 30 variables were revised or created, and the pilot will assess whether business processes across these organizations can support the new guidance with reasonable effort.
This document will guide the community’s efforts for years to come. If you would like to volunteer, we’re beginning the planning process and scoping out the evaluative measures for the pilots. We anticipate the pilots to begin in the fall. We could use your help making sure messages are complete, processed seamlessly, and received intact. More details will be shared soon. If interested, please email nssp@cdc.gov.
CDC Funding Recipients and Partnership Updates

Recipients of CDC NSSP funding for syndromic surveillance convened to share their experiences, challenges, problem-solving, and best practices. Community collaboration is strengthening public health and positioning surveillance activities to thrive in the years ahead.
NSSP Closes Out CoAg
On August 19–20, 2019, recipients of NSSP funding convened in Atlanta, Georgia, to close out the current (CDC-RFA-OE15-1502) 4-year cooperative agreement (CoAg) that ended August 31. The meeting title—Maintaining and Advancing in Epidemiology and Laboratory Capacity (ELC) for Prevention and Control of Emerging Infectious Diseases—conveyed the major administrative change of integrating future NSSP CoAg activities with the ELC’s Health Information Systems Capacity CoAg. By merging with the ELC CoAg, the NSSP team will spend less time administering the CoAg, leaving more time for technical support and program planning. NSSP also gains a strong, well-established infrastructure with the flexibility to fund more health jurisdictions and special projects.
The merger with the ELC CoAg should elevate the visibility of syndromic surveillance. Already, NSSP has increased its collaboration across other surveillance areas and encouraged integration of syndromic data with other systems.
Syndromic Surveillance in Action
A highlight of the close-out meeting, aside from sharing surveillance experiences with colleagues, was hearing presentations about ways in which NSSP funding had expanded state capacity. Some state health departments expanded capacity by hiring much needed epidemiological experts, whereas others created dashboards to support strategic alliances, enhanced their systems, or expanded their facility coverage and data quality. In future issues of NSSP Update, we plan to share some of these inspiring stories.
Across most health jurisdictions, the onboarding numbers reflected a significant increase in coverage (currently, 68% of the nation’s emergency departments [EDs] transmit data to NSSP). Connecticut, for example, has 100% of its EDs onboard. Nebraska experienced a 48% increase in the proportion of targeted EDs that submit production data, which translates into 88% of the population being represented. New York State (excluding New York City) has 88% of its EDs onboard. Massachusetts—an “NSSP Only” syndromic surveillance health jurisdiction—has onboarded 100% of its EDs and is now focusing on onboarding urgent care facilities.
Practitioners in all states aimed high with their performance goals. The list of state successes is long and impressive—too long to adequately cover here. Still, here’s a glimpse into how state commitment to syndromic surveillance translates into public health action:
- Connecticut implemented automated alerts and identified a mass overdose of K2. Connecticut monitors weather-related emergencies, hoarding-related disorders, chronic disease, and STDs—all the while looking at risk factors and geographic hot spots.
- Kansas is fast becoming a model for data sharing. About 20 local health departments in Kansas have data use agreements in place, with another 8 about to start. The Kansas team has also launched discussion with state hospital associations about data sharing.
- Missouri uses ESSENCE daily and has enhanced how it monitors special events (e.g., World Series, Presidential Debate in St. Louis, New Year’s holiday, and St. Louis Fair).
- Nevada uses ESSENCE to evaluate the impact of the legalization of marijuana.
- New Hampshire expanded its partnerships with state agencies to better support the State Drug Monitoring Initiative.
- New Jersey moved beyond using syndromic data for communicable disease into monitoring environmental and occupational exposures, opioid classifications, tick-related illness, and waterborne illness (new).
- New Mexico links syndromic data with other data sources, including weather data, and is exploring machine learning techniques to improve national biosurveillance.
- New York State added three drug-related syndromes to its surveillance system, initiated a Chief Complaint/Discharge Diagnosis (CC/DD) algorithm, and began using SaTScan supplemental surveillance methods to look at drug-related syndromes and communicable disease.
- North Dakota reviewed its data security policies and overcame some data confidentiality concerns to improve its data quality.
- Utah strengthened syndromic surveillance by selecting measures to improve onboarding, data quality, data flow, and representativeness. Utah is also helping staff at healthcare organizations communicate the impact of Promoting Interoperability programs to health information exchanges.
For more state successes, we encourage you to read NSSP Update and postings in the Knowledge Repository.
NSSP’s Future
Program Manager, NSSP
NSSP Program Manager Michael Coletta took the meeting attendees on a whirlwind journey of the program’s transformation, priming them for what’s ahead. He emphasized that step one is to maintain what we’ve built. The NSSP team will continue to enhance the data flow, BioSense Platform architecture, AMC controls, and, in partnership with community members, explore word embedding, machine learning, and new analytic methods. We also plan to pilot the new HL7 balloted Implementation Guide early in 2020. NSSP analysts will continue to look for opportunities to integrate new data sources. CDC will continue to support the NSSP CoP, and NSSP will work closely with the new CoAg partner to promote activities and identify opportunities to advance syndromic practice and science.
Capstone Meeting
Given so many community members were in Atlanta for the close-out meeting, NSSP used this opportunity to host a capstone meeting Wednesday, August 21, 2019, to share results from a recent series of regional data-sharing workshops. The workshops, conducted in partnership with the Council of State and Territorial Epidemiologists (CSTE), were designed for funding recipients in each of the U.S. Department of Health and Human Services’ 10 regions. The workshops were customized to meet participants’ surveillance objectives, maximizing the practical value of data sharing and, more importantly, setting a foundation for subsequent collaborative work.
Workshop facilitator Charlie Ishikawa of Kahuina Consulting shared his workshop take-aways by reminding everyone of the larger picture: How does SyS data sharing improve situational awareness? What are our common barriers? What is CDC’s vision for data sharing?
Ishikawa acknowledged the community members’ readiness to share syndromic data because the value added was clear and compelling. Ishikawa applauded the rigor with which community members tackle associated challenges, noting that motivation is NOT a barrier. In fact, the community is highly motivated. Most barriers being experienced are inherent to systems of jurisdictional policy and public health governance. He also acknowledged that competency using the BioSense Platform tools has increased, and, although it could be stronger, competency is not a barrier to data sharing.
Ishikawa concluded by identifying the need to formulate the strategic next steps for an action plan to reinforce SyS data-sharing collaborations. Look for more on data sharing in upcoming issues of NSSP Update.
ESSENCE QUERIES
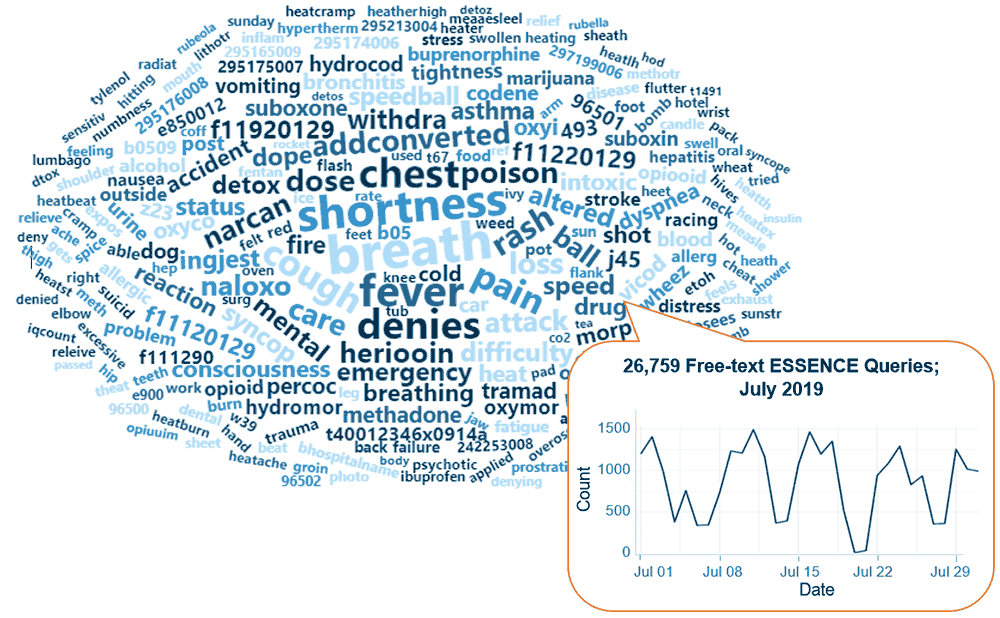
Query Terms from July 2019
A key feature of NSSP–ESSENCE is its ability to use free-text definitions across different fields to generate custom queries. The SyS community can use this feature to develop and refine category definitions and to identify and respond to public health events that may not be apparent in established categorical definitions (i.e., syndromes, subsyndromes, and Chief Complaint and Discharge Diagnosis [CCDD] categories).
This word cloud summarizes free-text and coded values that epidemiologists and data analysts queried in ESSENCE during July 2019. This word cloud was generated by using the Wordcloud2 package in RStudio. The relative size of the terms suggests the frequency of use in a query. NSSP super administrators can query ESSENCE usage logs to learn what queries are run nationwide and how long they take to complete.
Practice
Collaborations Accelerate SyS Practice

Collaborations initiated by the NSSP CoP Steering Committee (and its workgroups and committees) and health departments partnering with CDC programs are improving how syndromic surveillance gets done. The list of collaborations that follows, while not exhaustive, summarizes ongoing efforts to develop and improve syndrome definitions, conduct pilot tests and exercises, and improve processes to achieve public health goals more efficiently.
Here are the collaborations rapidly moving us into 2020:
Three-month Outlook:
- Implement syndrome definition for medication refill for during and after a public health disaster. JUST RELEASED! Check out the syndrome in ESSENCE.
- Develop a sexual violence v3 definition.
- Collaborator: CDC’s Division of Violence Prevention
- Scheduled for early fall 2019
- Develop syndrome definition for pregnancy, delivery, and miscarriage.
- Collaborator: CDC’s Maternal and Infant Health/Maternal and Infant Health Branch (MIHB)
- Scheduled for early fall 2019
- Develop syndrome definition for alcohol-involved emergency department visits.
- Collaborator: Opioid and Overdose Team
- Scheduled for early fall 2019
- Develop syndrome definition for injecting drug users (IDUs) and endocarditis (infection of inner lining of heart chamber and valves).
- Collaborator: CDC’s National Center for Emerging and Zoonotic Infectious Diseases, Epidemiology Research and Innovations Branch
- Scheduled for fall 2019
- Develop syndrome definition for motor vehicle collision (e-scooters). NSSP is part of the Transportation Safety team, e-scooter working group.

Want help meeting surveillance goals?
Collaborate.
Engage NSSP, CDC programs, or health jurisdictions about special projects by emailing
nssp@cdc.govDevelop syndrome definition for child abuse and neglect.
- Collaborator: CDC’s Division of Violence Prevention
- Develop syndrome definition for child sexual abuse.
- Collaborator: CDC’s Division of Violence Prevention
- Develop syndrome definition for sex trafficking.
- Collaborator: CDC’s Division of Violence Prevention
- Develop syndrome definition for teen dating violence.
- Collaborator: CDC’s Division of Violence Prevention
- Develop syndrome definition for homeless populations.
- Collaborator: CDC’s Division of Violence Prevention
2019–2020:
- NSSP is participating in a functional exercise to gauge how well the community shares information and data during a public health event. This exercise will evaluate response speed; technical needs and delivery options; potential local, state, and federal collaborations; and integration of emergency preparedness and management personnel.
- Collaborator: NSSP CoP SyS Public Health Emergency Preparedness Response and Recovery (SPHERR)
- The NSSP team is working with the community and CDC’s National Center for Injury Prevention and Control (the Injury Center) to re-categorize sexual violence syndromes into broad and narrow concepts to facilitate better use of syndromic data.
- NSSP is working with CDC’s National Center for Chronic Disease Prevention and Health Promotion, Division of Cancer Prevention and Control, on a special-interest project though the Prevention Research Centers Program: “Improving Cancer Survivor Treatment and Outcomes by Ensuring Appropriate Emergency/Acute Care Treatment.” Work will begin fall 2019 and extend through 2020.
- The NSSP team is developing a syndrome definition for ketoacidiosis (a life-threatening but avoidable complication of diabetes when high levels of blood acids, ketones, are produced), specifically searching the discharge diagnosis field for ICD-9/10 codes.
- Collaborator: Division of Diabetes Translation with their Surveillance Team
Our thanks to Health Scientist Nimi Idaikkadar (Center for Surveillance, Epidemiology, and Laboratory Services, Division of Health Informatics and Surveillance) for this update.
Free-text Coding in NSSP–ESSENCE
This is the fifth in a series of articles about how to write ESSENCE free-text queries.
“Part 1. Wildcards” can be found here.
“Part 2. Underscores_and Brackets [ ]” can be found here.
“Part 3. Inclusionary Terms” can be found here.
“Part 4. Exclusion Terms and Parentheses” can be found here.
Introduction

The search criteria for ESSENCE free-text queries are built around Boolean logical operators and regular expressions. Free-text queries are not case-sensitive and may contain “^” for wildcards; “,” for multiple entries; “ISBLANK” to look for blanks; “ISNULL” to look for nulls; “[COMMA]” to look for commas; and operators “and,” “or,”, “andnot,” and parentheses “()” to define order and grouping. This series will cover all these topics in-depth.
Free-text queries are what makes syndromic surveillance practice, particularly practice using NSSP–ESSENCE, adaptable to different data sources and types. By using free-text queries, analysts and epidemiologists can exercise a high level of customization. They can quickly code free-text queries and rapidly respond to outbreaks, disasters, and events that unfold. Such capabilities empower users to customize queries to fit their level of data, ensuring accurate results.
Free-text coding in ESSENCE, which is accessible to all users, follows distinct patterns. Learning to read these patterns allows users to take queries from many places and repurpose them to suit their unique needs. Syndromic surveillance depends heavily on sharing methods, and practitioners must understand the language.
Part 5. A “Starter” Fall-related Injury Query and Examples of Complex Queries
Parts 1 through 4 of this series cover basic and complex query notation, including the use of carets; underscores and brackets; “AND”, “OR”, and “ANDNOT” statements; and the parentheses that can be used to group these statements together.
A “Starter” Fall-related Injury Query
So far, this series has referenced a sample set of hypothetical Chief Complaints (CCs) and a desire to create a query for fall-related injuries. Now that we have all the query language at our fingertips, here’s how I would develop a query that includes the intended visits and excludes the false positive CCs.
Let’s assume the following Chief Complaints (used in Parts 1–4) and a desire to create a query for fall-related injuries.
- Fall
- Fell getting out of car
- Left arm injury; Fall
- Falling out with friends; Suicidal
- Feels crestfallen
- Patient brought in after falling on face
- Fall; Left wrist injury
- Feels congested; Allergies
You may reasonably assume the boldfaced CCs 1, 2, 3, 6, and 7 are the intended cases and 4, 5, and 8 are false positives.
Here’s a table that describes what we’ve learned about fall-related queries:
| Fall-related Query Examples | |
|---|---|
| Code | Description |
|
|
^Fall^,OR,^Fell^ |
This query picks up CCs 1, 2, 3, 4, 5, 6, and 7. This captures all intended visits but also captures the false positive terms for “Falling out with friends” and “Feeling crestfallen.” |
|
|
^Fall^,ANDNOT,^Crestfallen^,OR,^Fell^ |
We can negate the false positive term directly. This returns CCs 1, 2, 3, 4, 6, and 7. |
^ Fall^,OR,Fall^,or,Fell^ |
Or, we can be specific with our inclusion term. This code specifies that the term starts with the word (or CC) and returns CCs 1, 2, 3, 4, 6, and 7. |
|
|
(,^Fall^,ANDNOT,^Crestfallen^,),AND, (,^Injur^,OR,^Face^,or,^Wrist^,or,^Ankle^ ,or,^Broke^,or,^Hurt^,or,^Lacer^,or,^Hit^ ,or,^Head^,),OR,^Fell^ |
We can require that “Fall” coincide with an injury term. The code shown returns CCs 3, 6, and 7. This excludes CC 4 but also excludes the likely true positive CCs 1 and 2. Also, this query depends heavily on the user’s ability to cover all injury descriptions that can result from a fall, which could get prohibitively long. |
^Fall^,ANDNOT,(,^Crestfallen^,OR,^Falling out with^,),OR,^Fell^ |
The query can negate the text string specific to CC 4. The more specific your negation term becomes, the more likely it won’t apply across broad real-life data or negate similar false positives. Coding very specific negations can be used to negate especially troublesome false positives. The code to the left has a very specific negation to exclude troublesome CC 4 and returns CCs 1, 2, 3, 6 and 7. |
^Fall^,ANDNOT, (,^Crestfallen^,OR,^Suicid^,),OR,^Fell^ |
The query can negate based on how CC 4 relates to suicide ideation. However, that raises the question of how many times fall-related injuries occur alongside mentions of suicide ideation. This query returns CCs 1, 2, 3, 6, and 7. |
So, that makes our final query on our hypothetical CCs either
^Fall^,ANDNOT,(,^Crestfallen^,OR,^Falling out with^,),OR,^Fell^
Or . . .
^Fall^,ANDNOT,(,^Crestfallen^,OR,^Suicid^,),OR,^Fell^
Both queries are concise and accurate, but will they work with real-life data? The Chief Complaint Query Validation (CCQV) dataset in NSSP–ESSENCE is the logical (and best) place to start when creating a query that will be used nationwide. But for most situations, a user’s local or regional data will suffice. For the next steps, we will use just the query ^Fall^,ANDNOT,(,^Crestfallen^,OR,^Falling out with^,),OR,^Fell^.
Fall-related injuries are common in emergency department data. The existing ESSENCE fall subsyndrome pulls these visits reliably, but we can also run our new syndrome and see how it performs on real-life data. Running the above query in CCQV data where HasBeenE=Yes (roughly limiting it to emergency care) returns just under 80,000 visits per week. Moving forward to see how our query works with a broader dataset, we will focus on just MMWR week 22, 2019. Contrary to this example that uses a single week’s data, anyone validating a query should do so across multiple weeks to ensure that coverage includes a broad range of CC variations.
The official return of our query for MMWR week 22 is 79,604 visits. We can let ESSENCE take a random sample of these visits in the Data Details page to review. After reviewing these visits, our query seems to be performing quite well. False positives immediately noted are: Chief Complaints about assorted medical devices that “fall out” and items that fall or fell onto the patient.
Because we are looking for false positives, we can further refine the data we have to review by removing likely true positive visits. One method is to remove those that have relevant CCs and also have Discharge Diagnoses (DDs) related to our intended records. We can do this by putting the original query in parentheses and then linking to relevant DD codes with an “ANDNOT” statement. Although the methodology for picking DD codes is beyond the scope of this article, a quick ICD10 negation of visits coded with a W00-W19 ICD10 code covering slipping, tripping, stumbling, and falls will allow us to remove many true positives.
Our code to remove DD-positive visits, which we’ll run in the CCDD field, is shown below. Note the use of brackets to cover this wide range of “W” codes with a single free-text term:
(,^Fall^,ANDNOT,(,^Crestfallen^,OR,^Falling out with^,),OR,^Fell^,),ANDNOT,(,^[;/ ]W[01][0-9]^,)
This query returns 53,676 visits for MMWR week 22. A review of the returns shows, once again, only a few false positives.
Code can be written to further isolate potential false positives, making it easier for users to quickly review and look for trends in terms to negate in the final query. For example, if a user has some knowledge of medical devices that can potentially “fall out,” these devices can be listed in a query and thereby isolated. To do this, group the medical device terms within parentheses and then use an “AND” statement to link the parenthetical statement to the original query before the “ANDNOT” statement of diagnosis codes.
(,(,^Fall ^,ANDNOT,(,^Crestfallen^,OR,^Falling out with^,),OR,^Fell^,),AND,(,^Tube^,OR,^Cath^,OR,^Monitor^,OR,^Stitch^,),),ANDNOT,(,^[;/ ]W[01][0-9]^,)
Note: This query returns 200 visits in MMWR week 22. Of these, at least 20 are visits where the fall event dislodged the medical device and caused it to “Fall out.” “ANDNOT” statements looking for false positives can have surprising and unintended consequences.
Try it out!
Try to negate cases where an item falls/fell onto the patient. With each negation, think about its consequences. For example, dressers can fall on people, but people can also fall off dressers.
Will the ANDNOT string (,^Dresser f[ea]ll^,) work for this? See what you’re negating with the code
(,(,^Fall^,ANDNOT,(,^Crestfallen^,OR,^Falling out with^,),OR,^Fell^,),AND,(,^Dresser f[ea]ll^,),),ANDNOT,(,^[;/
Complex Query Examples
In next month’s free-text coding article, we will present a rough final query for fall-related injuries and then break it apart. For now, here are some complex queries and different free-text language techniques they use well.
Notice in the highlighted portion of the new CDC Medication Refill v1 CCDD Category how an “ANDNOT” statement (red) is nested within the first group of inclusion terms (green), which, in turn, are linked to another group of inclusion terms (gold). By nesting the “ANDNOT” statement within the inclusion terms, the coder can be specific with the negations. Here, only the specific false positives pulled by the ^o2^ term are negated. This way, you only apply negations to terms where needed.
(,(,^tank^,or,^oxygen^,or,^o2^,ANDNOT,(,^O2[0-9]^,),or,^medication^,or,^script^,or,^pill^,or,^prescription^,or,^medicine^,),AND,(,^refil^,or,^supplemental^,or,^empty^,or,^need^,or,^out of^,or,^request^,),),or,(,^[;/ ]z76.0^,or,^[;/ ]z760^,or,^[;/ ]V68.1^or,^[;/ ]V681^,or,^[;/ ]182918009^,)
Shown below is the “Rabies and Animal Bites – Florida Department of Health” query, which is posted in the Syndromic Definition Library (SDL) in the Knowledge Repository (https://surveillancerepository.org/rabies-and-animal-bites-florida-department-health):
(,^rabie^,or,^rabbie^,or,^bitten^,or,^bite^,or,bit ^,or,^ bit,or,^ bit ^,),andnot,(,^bug^,or,^ant^,or,^child^,or,^insect^,or,^flea^,or,^person^,or,^human^,or,^snake^,or,^caterpil^,or,^pygmy rattler^,or,^something^,or,^spider^,or,^wasp^,or,^mosquito^,or,^tick^,or,^fish^,or,^recluse^,)
A notable feature of this rabies query is the concept that instead of being more specific with inclusion terms, coders chose to exclude visits where the biter was mentioned AND is not a likely carrier of rabies. This works great since a query for ^Bite^ alone will pull in insects, reptiles, amphibians, and children and a query for ^Bite^,AND,(,^Cat^,or,^Dog^,or,^etc…….^,) will exclude unattributed bites and fail to bring them to an epidemiologist or other decision-maker’s attention.
The CSTE Cold-Related Illness query posted to the SDL serves as another excellent example of key query methods (https://surveillancerepository.org/cold-related-illness-cste-january-2019).
Notice how the ICD9 code starting with 991 (highlighted in green) uses the bracketed list [;/ ] at the start to ensure the number segment starts the code, followed by an underscore to specify anything can fall in that place, but that place must be filled, and then ends with another bracketed list [;/ ] to specify the end of the code. The bracket at the end ensures that numbers longer than 4 digits won’t be pulled by this query.
This query also does a nice job negating historic frostbite and cold exposure injuries (highlighted in red) since frostbite patients are likely candidates for repeat ED visits for bandage changes, infection, swelling, and other issues. Notice in particular the use of …^[TX]__.__XD^,or,^[TX]____XD^… to negate any injury or external cause code that has a “D” at the end indicating subsequent encounters.
(,^cold exp^,or,^cold-exp^,or,^env exp^,or,^environmental exp^,or,^exp to env^,or,^exposure to env^,or,^exp to cold^,or,^exposure to cold^,or,^cold exposure^,or,^coldexposue^,or,^out in the cold^,or,^f[ro][ro]stbit^,or,^f[ro][ro]st bit^,or,^f[ro][ro]stnip^,or,^f[ro][ro]st nip^,or,^f[ro][ro]st ni^,or,^hypothe^,),or,(,^froze^,and,(,^hand^,or,^finger^,or,^thumb^,),),ANDNOT,(,^Meat^,or,^burger^,or,^pork^,or,^wart^,or,^growth^,or,^food^,or,^knife^,or,^shoulder^,or,^turkey^,or,^knee^,),or,(,^froze^,and,(,^Feet^,or,^foot^,or,^toe^,),ANDNOT,(,^Meat^,or,^burger^,or,^pork^,or,^wart^,or,^growth^,or,^food^,or,^knife^,or,^shoulder^,or,^turkey^,or,^knee^,),),or,(,^[;/ ]x31^,or,^[;/ ]t68^,or,^[;/ ]t69^,or,^[;/ ]t33^,or,^[;/ ]t34^,or,^[;/ ]991._[;/ ]^,or,^[;/ ]991_[;/ ]^,or,^[;/ ]E901.[089]^,or,^[;/ ]E988.3^,or,^[;/ ]E901[089]^,or,^[;/ ]E9883^,),andnot,(,^recheck^,or^re-check^,or,^history of frostbite^,or,^historyfrostbite^,or,^dressingchang^,or,^chest^,or,^congestion^,or,^cough^,or, ^infection^,or,^[;/ ]w93^,or,^[;/]r68.0^,or,^[;/]r680^,or,^[;/ ]e901.1^,or,^[;/ ]780.65^,or,^[;/ ]e9011^,or,^[;/ ]78065[;/ ]^,or,^[TX]__.__XD^,or,^[TX]____XD^,)
We thank Senior Data Analyst Zachary Stein for volunteering to write a series of articles about free-text coding. Stein, formerly with the Kansas Department of Environment and Health, does epidemiologic work to support NSSP efforts. Stein is an active participant in the NSSP CoP. He initially wrote about free-text coding as an entry on the NSSP CoP Syndrome Definition Committee forum. The forum generated considerable interest, inspiring this series. Stein acknowledges input provided by others who contributed to the forum post.
Spanish Words in the Chief Complaint and Discharge Diagnosis Fields
Spanish words have been detected in the chief complaint and discharge diagnosis fields. The CCDD field is helpful for detecting Spanish words that indicate healthcare services are being used or that people say when describing symptoms to a healthcare provider.
The top Spanish words used are fiebre, y, tos, dolor, de, gripe, gripa, and vomito. Currently, Spanish words are not being binned into English word syndromes. By binning these Spanish words, the quality of information contained in our syndromes will improve.
Data Quality Corner
Introduction to New NSSP–ESSENCE Data Quality Filters
For some time, the community of practice has tried to improve (and maintain) data quality. However, the completeness of reporting some variables fundamental to syndromic surveillance practice, such as chief complaint and discharge diagnosis, can vary across and within sites over time and complicate how surveillance data are interpreted.
Such variance among data explains why some community of practice members are cautious when they compare syndromic trends across sites. For example, an analysis of NSSP–ESSENCE emergency department encounters nationwide showed, on average, that the daily percentage of discharge diagnoses considered informative (i.e., are not null or do not simply include terms like “unknown”) was 45% in 2016, 54% in 2017, 65% in 2018, and 70% in 2019 (figure 1). This is an improvement of about 56% from 2016 to 2019. Yet, if a user’s multiyear query relies on the discharge diagnosis for results, differentiating the surveillance trend from the influence of longer term improvements in data quality is still complicated.
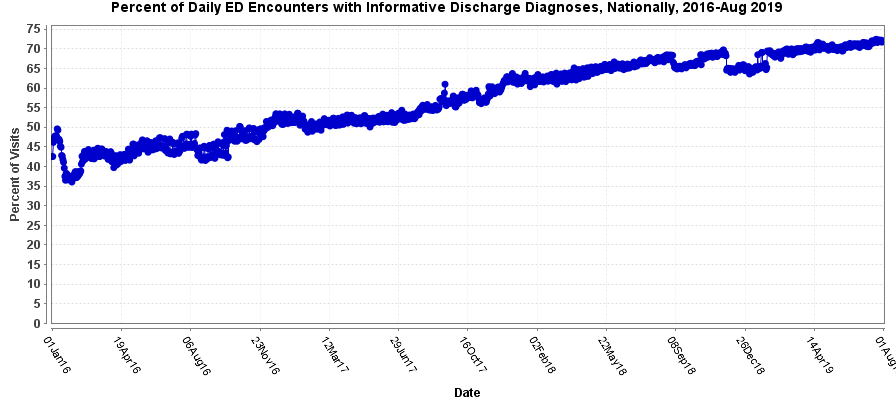
Figure 1. Percent of Daily Emergency Department Encounters with Informative Discharge Diagnoses
New functionality has been added to NSSP–ESSENCE that makes controlling for this variation easier. In the Query Portal interface, within the list of Available Query Fields, users will notice a set of variables in the Data Quality Filters folder (figure 2).
Figure 2. Menu Options in NSSP–ESSENCE
The options for each variable are binary, Yes/No selections. For example, if a user selects Discharge Diagnosis Informative (DDI) equals Yes, then the query will only include encounters with an informative discharge diagnosis. Additionally, if the query is run as a percentage of all visits, then the numerator and denominator will only include encounters where the DDI is Yes, which helps control for the influence of improvements in discharge diagnosis completeness on surveillance trends. By requiring DDI equals Yes, keep in mind that encounters with chief complaint text that could have matched your query might be excluded.
Within the next few weeks (see staging NSSP–ESSENCE for a sneak peek), additional filters will be added to the Data Quality Filters folder in NSSP–ESSENCE that will give users more control over surveillance trends based on data quality at the hospital versus encounter level, as described above. Check out the June 19, 2019, ESSENCE Q &A number 5 webinar (~minute 51:27) for a discussion of this and other features.
Let’s Work Together to Keep Your MFT Up to Date!

Last month, NSSP began alerting sites when facilities had failed to send data for 30 days AND when data had not been sent for at least 90 days. These alerts are part of an ongoing effort to improve data quality and make sure your site’s Master Facility Table (MFT) is current.So, what happens when a facility has not sent data for 90 days? Unless the site administrator has provided an explanation, the facility will be deactivated* and the MFT updated:
- Facility Status will be updated to “Inactive,”
- Review Reason of “Status change from active,”
- Review Status of “Pending Onboarding (OB) Approval,” and
- Addition to the Onboarding comment field that states: “This facility was deactivated per the 90-day policy.”
* NSSP onboarding team members and site inspectors will consult one another before approving a change to facility status.
ALWAYS, sites will be informed before a facility is deactivated. NSSP will alert individuals whose names are checked beside “Onboarding Communications” on the Access & Management Center (AMC) User Profile page (see the AMC Quick Start Guide, Section 4: Manage User Profile – Site-specific Communications).
—Sunday, September 1—
Deactivation of facilities that have not transmitted data for 90 or more days
—Monday, September 2—
Deactivation alerts issued
—Friday, September 20—
Deadline for completing facility review with onboarding team
—Monday, September 23—
Facility deactivation
Here’s what to do if you receive an alert:
First, review the identified facilities for accuracy. Before completing the facility deactivation, the NSSP onboarding team and site inspectors will contact site administrators to determine whether the facilities should be “Inactive” or if ongoing work will allow data transmittal to resume. If facilities are inactive, the onboarding team will work with the site administrator to update the MFT facility status value to “Inactive” and enter the reason in the comment field. The deadline for completing the facility review with the onboarding team is Friday, September 20, 2019.
Always add a note in the comments field—especially in the following situations:
- If a facility has not sent data due to a closure, please enter “Inactive due to Closure” and closure date.
- If a facility has not sent data due to a merger, please enter “Inactive due to Merger,” the facility it merged with, and the date the data began arriving in the new feed.
As a best practice . . . Keep your MFT current by reviewing it each month. If you know of facilities that should be changed to Inactive or Onboarding, make the entry before you receive the first deactivation alert.
90-Day Deactivation Alerts:
On the second day of each month, NSSP will alert sites of facilities that have not sent data for 30 days and are at risk of deactivation:
| Facility Name | C_BioSense_Facility_ID | FacilityID_UUID | Most Recent C_Visit_Date | Number of Days since Last Visit |
|---|---|---|---|---|
| <Facility Name> | ## | ## | <YYYY-MM-DD> | ## |
On the second day of each month, NSSP will notify sites of the facilities deactivated the previous month:
| Facility Name | C_BioSense_Facility_ID | FacilityID_UUID | Most Recent C_Visit_Date | Number of Days since Last Visit | Date_Deactivated |
|---|---|---|---|---|---|
| <Facility Name> | ## | ## | <YYYY-MM-DD> | ## | <YYYY-MM-DD> |
NSSP will email alerts when facilities fail to send data for visits occurring within 90 days:
| Facility Name | C_BioSense_Facility_ID | FacilityID_UUID | Most Recent C_Visit_Date | Number of Days since Last Visit | Date_Deactivated |
|---|---|---|---|---|---|
| <Facility Name> | ## | ## | <YYYY-MM-DD> | ## | <YYYY-MM-DD> |
Spotlight on Syndromic Surveillance Practice
Guidance for Implementing Cold-related Illness Syndromic Surveillance in Public Health Practice
Cold-related Illness Query

Check out the cold-related query in the NSSP CoP Knowledge Repository. Share your findings or make suggestions by emailing us at nssp@cdc.gov.
Despite the nation’s current record-high temperatures, it’s not too early to prepare for wintertime surveillance. As you plan, refer to this guidance document developed by the Council of State and Territorial Epidemiologists Heat and Cold Workgroup in collaboration with data analysts and epidemiologists from the NSSP–CoP Syndrome Definition Committee. The guide contains a novel query for cold-related illness based on syndrome definitions used by five health jurisdictions. (Note. This cold-related query is available in NSSP–ESSENCE.)
The guide emphasizes the process for developing a query—identifying data sources, analysis, and validation using local data. The guide discusses how often data should be run and analyzed, which largely depends on the region in which the public health department is located, and how data will inform public health action.
Within the guide is the Kansas Cold Weather Toolkit, created by the Kansas Extreme Weather Workgroup and adapted (with permission) from a toolkit developed by the Wisconsin Department of Health Services. Also included is the Wisconsin Winter Weather Toolkit—A planning toolkit for public health and emergency response professionals.
Both toolkits include winter weather alerts and terminology, health effects and risks, cold-weather preparation and travel, indoor and outdoor safety, information about vulnerable populations, and links to other state resources. Each contains a message map that health communicators can use when they craft key messages for media outlets or the general public.
The Cold-related Illness Query and both toolkits are excellent planning tools that can improve a wintertime communications program. Guidance has been posted in the NSSP CoP Knowledge Repository.
Program
Technical Updates
AMC Update Scheduled for Fall 2019
This month, NSSP is deploying Data Quality Dashboards on the BioSense Platform. These dashboards will help site administrators investigate data quality issues through interactive, filterable data visualizations depicting data processing, timeliness, completeness, and validity metrics. The dashboards can be accessed via WebLink. Later in the fall when the Access & Management Center (AMC) is released, the Data Quality Dashboards will be available via the AMC Reports page.
More improvements to the AMC are underway, including new functionality for creating data access rules that will streamline rule creation and editing. Additionally, downloadable Data Access Reports will be available to site administrators through the AMC Reports page.
DataMart Table Content Change
File_Name was added to the Processed and Exception tables. No longer will BioSense Platform users need to join the Processed tables to Raw tables to get the file name for their records, simplifying query performance to run faster.
File Server Change
NSSP has implemented a new policy for the BioSense Platform that limits the maximum File_Server space, per user, to 500 gigabytes. The NSSP team will notify users if they are close to this new limit and can suggest ways to optimize space, if needed.
Current Month and Upcoming Events
| September 4 | Data Validation Conference Call, 3:00–4:00 PM ET |
| September 17 | NSSP CoP Call; 3:00–4:30 PM ET; Earthquake Surveillance: Shaking Things Up with SyS |
| September 20 | Scheduled vendor patches in staging environment: 6:00–10:00 AM ET |
| September 22 | Scheduled vendor patches in production environment: 6:00–10:00 AM ET |
Last Month’s Technical Assistance
| August 7 | Held Data Validation Conference Call |
| August 20 | Applied vendor patches in staging environment |
| August 22 | Applied vendor patches in production environment |
NSSP Participation
A total of 58 sites from 46 states and the District of Columbia participate in NSSP. Currently, there are 4,478 facilities, including 3,021 emergency departments (EDs), actively contributing data to the NSSP BioSense Platform. Data from these EDs cover about 68% of all ED visits in the country.
NSSP publishes data for ED visit coverage each quarter. These data and the coverage map shown below were updated July 2019. The calculation method is described in the December 2018 issue of NSSP Update.
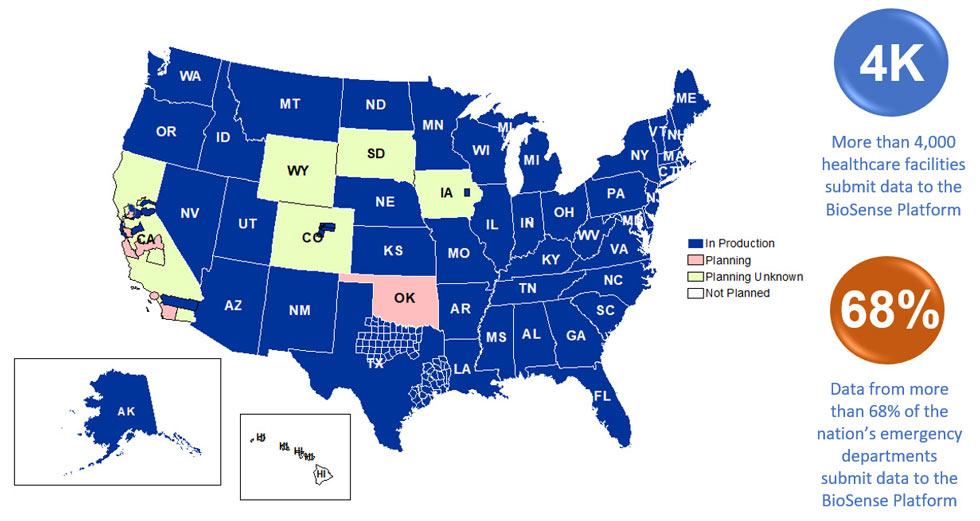
Definitions: NSSP consolidates facilities that provide data under a single data administrative authority called a site administrator. These facilities and single-site administrator constitute a site.

