February 2022
February 15, 2022, 3:00–4:00 PM ET: eSHARE webinar: “Where Are We Now? Using Immunization Information Systems to Enhance Disease Surveillance and Investigations During the Response.”
June 19–23, 2022: Council of State and Territorial Epidemiologists (CSTE) Annual Conference, Louisville, Kentucky. Learn more

Now available: new and updated NNDSS case definitions for 2022
The CDC National Notifiable Diseases Surveillance System (NNDSS) team has posted the updated case definitions for Chlamydia trachomatis infection, Lyme disease, viral hemorrhagic fever, and acute flaccid myelitis. The team also released the newly created case definition for standardized surveillance of alpha-gal syndrome. The updated COVID-19 case definition was implemented on September 1, 2021.
Visit the January 18, 2022, eSHARE webinar archive or read the 2022 Letter to State and Territorial Epidemiologists to learn more.
New annual data reconciliation functionality coming in 2022
The CDC NNDSS team is working to add new features to the Message Validation, Processing, and Provisioning System (MVPS) portal for the 2021 annual data reconciliation process. These improvements will streamline the reconciliation process by giving jurisdictions direct control over their reconciliation activities and automating steps that were previously handled by email. Jurisdiction users will be able to use MVPS to:
- access reconciliation reports on demand,
- lock their annual datasets when the data are ready for approval, and
- provide final sign-off of the locked data by the STD Manager and State or Territorial Epidemiologist.
The NNDSS team will provide more information and training on these upcoming features closer to release.

Coming soon: new format for 2022 weekly tables on Data.CDC.gov
NNDSS is working to transition weekly data publication for 2022 data on Data.CDC.gov to a single-table format. This change will consolidate 42 weekly NNDSS tables on Data.cdc.gov into one table that includes all conditions and data published in the 2022 NNDSS Weekly WONDER tables. This change will make it easier for data users to extract the NNDSS data.
The NNDSS team is working to finalize the timeline for this change; stay tuned for future updates! If you need technical support with Data.CDC.gov, visit the Socrata helpdesk.
The next eSHARE webinar, “Where Are We Now? Using Immunization Information Systems to Enhance Disease Surveillance and Investigations During the Response,” is scheduled for February 15, 2022, 3:00–4:00 PM ET.
To join a webinar, please see your Outlook invitation or contact the CDC Electronic Data Exchange mailbox at edx@cdc.gov for login information with the subject line “eSHARE invitation.”
NNDSS has released the 2022 annual Letter to State and Territorial Epidemiologists. The letter summarizes changes to NNDSS based upon position statements approved by CSTE in 2021 and other relevant updates.
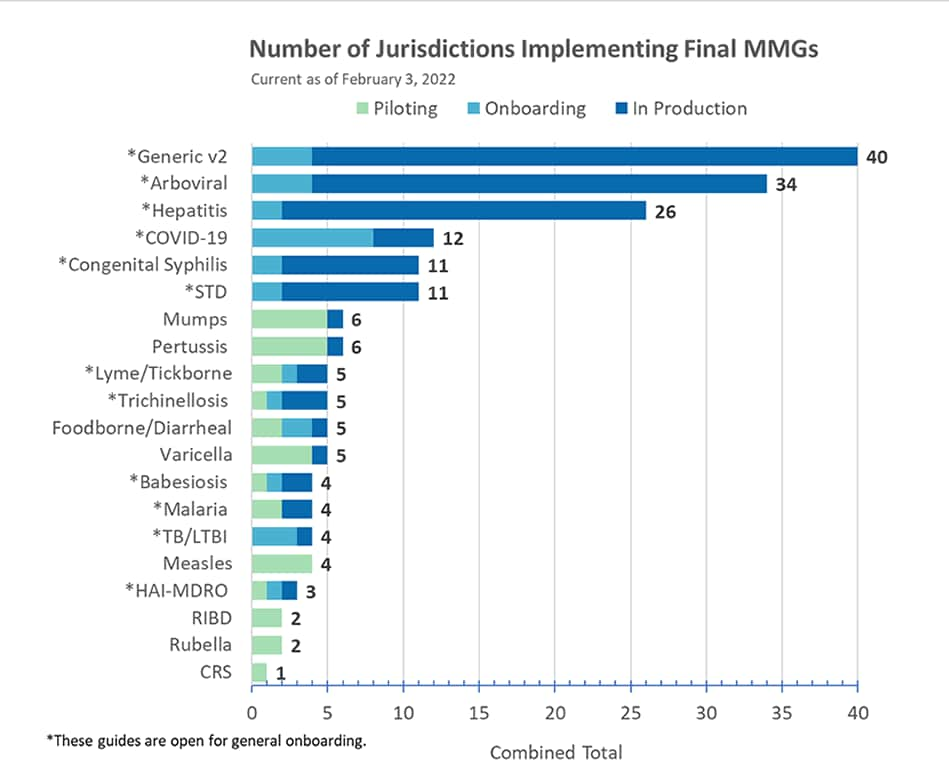
Arboviral Message Mapping Guide (MMG)
CDC has posted an updated Arboviral Disease Reporting Surveillance Guide to the NNDSS website under current “Artifacts.”
COVID-19 MMG
The PHIN Vocabulary Access and Distribution System (VADS) COVID-19 Case Notification View version 11 is now available. It includes an additional value for both Vaccine Type (NND) and Manufacturers of vaccines (MVX).
Healthcare-Associated Infections, Multi-Drug Resistant Organisms (HAI-MDRO) MMG
HAI-MDRO is now open for general onboarding. HAI-MDRO MMG version 1.0.2 contains formatting updates and the most up-to-date value set for International Destination(s) of Recent Travel (82764-2) and Specimen Type (66746-9) for both conditions in the guide. All value sets associated with this guide are in the PHIN VADS HAI MDRO Case Notification View version 2.
If you are interested in joining a cohort or onboarding an MMG, please email the CDC Electronic Data Exchange mailbox at edx@cdc.gov.
Closer Look: Processing and publication timelines for case notification data
This article provides an overview of the processing and publication timelines for case notification data in MVPS and the weekly NNDSS tables. If you have questions or need assistance, send an email to edx@cdc.gov with the subject line “MVPS Data Monitoring.”

How long does it take for my data to appear in MVPS after I send it to CDC?
When your jurisdiction submits case notifications to CDC, it may take time for MVPS to process the data and display them in the portal. The amount of time needed for processing depends on the data source. Additionally, there may be occasional delays in processing due to large volumes of data sent at one time, system maintenance, or unforeseen technical issues.
- Generic v2-based HL7 messages are processed throughout the day and are viewable in MVPS in approximately 1–2 hours after they are received.
- Legacy HL7 messages are processed each evening. Messages submitted by 6:00 PM ET will be viewable by approximately 9:30 PM ET. Legacy HL7 messages received after 6:00 PM ET will be processed into MVPS the next evening.
- National Electronic Telecommunications System for Surveillance (NETSS) messages are processed throughout the day and are viewable in MVPS in approximately 1–2 hours after they are received. Processing of NETSS data is paused between 8:00 AM ET and 12:00 PM ET on Wednesdays to support data processing for the published weekly NNDSS tables.
- National Electronic Disease Surveillance System Base System (NBS) Master Messages are processed each evening. Messages submitted by 8:00 PM ET will be viewable by approximately 1:30 AM ET. NBS Master Messages received after 8:00 PM ET will be processed into MVPS the next evening.
When do I need to submit my data to ensure publication in the next weekly NNDSS tables?
NNDSS publishes weekly case counts through CDC WONDER and data.cdc.gov. Case notifications should be received by 6:00 PM ET on Tuesdays to allow enough time for processing of new case notifications and updates for inclusion in the weekly WONDER and data.cdc.gov publication.