Newborn Screening Is a Lifesaver

Learn more about a new test to help babies with rare genetic disease get faster treatment.
Liz and Don Carter enjoyed their annual family summer vacation in Myrtle Beach, S.C., in 2018 with their two sons, ages 5 and 2. The boys thrilled to the magic and excitement of the ocean. They laughed, splashed in the water, and squished sand between their toes.
But the happy scene changed on the drive home. Elliott, the 2-year-old, became ill and started vomiting. He appeared to have a typical virus, but Liz Carter began to worry after a few days went by with no improvement. She took him to the children’s emergency room, where the doctor seemed to chalk up her son’s illness to a virus that would pass soon.
Carter insisted that her son be given intravenous fluids, and the doctor eventually obliged, then sent them home. Watching Elliott as he slept that night, she had an uneasy feeling something wasn’t right. She loaded him into the car again and headed back to the emergency room.
“I said we weren’t leaving until they ran every test possible to find out what was going on,” Carter remembers. A different doctor on call seemed empathetic. He assured her, “Don’t worry, we will get to the bottom of this,” and started asking questions.

Liz, Grayson, Don, and Elliott Carter share precious time together.
(Photo courtesy of Liz Carter)
Doctors soon determined that Elliott was having unexplained seizures and placed him in a medically induced coma to give his brain a chance to rest. They believed the seizures had resulted from extensive blood clotting in his brain—which also had led to a stroke. “We need you to know how serious this is. We don’t expect to lose Elliott to this, but it’s possible,” they warned the Carters.
Ten days after Elliott entered the Intensive Care Unit, the doctors found what had caused the blood clotting: a rare genetic disease called homocystinuria, or HCU.
HCU is known as an inborn error of metabolism. It is a genetic disorder where an enzyme problem interferes with normal metabolism. Infants born with HCU cannot process certain amino acids properly. The build-up of one of those amino acids, homocysteine, is toxic, and can lead to serious complications if not treated early. These include eye, skeleton, and blood vessel problems, as well as intellectual disabilities.
Although most babies are tested for HCU at birth, the current test for it is challenging and needs improvement.
“We made the false assumption that because his newborn screening tests were within normal range, Elliott did not have a disorder that was screened for,” recalls Liz Carter. “Unfortunately, we now know that his HCU was missed in newborn screening. Had we known at birth that Elliott had this serious but treatable condition, we could have started treating him.”
Scientists have been working on a better solution.
New Test Could Help Newborns with HCU Get Faster Treatment
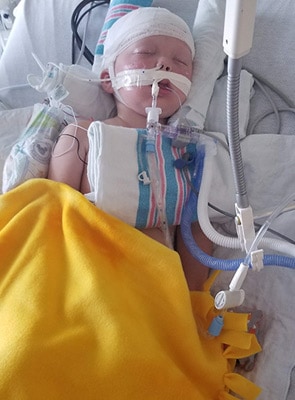
Baby Elliott in intensive care
(Photo courtesy of Liz Carter)
Thanks to researchers at the Centers for Disease Control and Prevention (CDC), a new way to screen for HCU in newborns will soon be available.
The new test measures homocysteine levels directly. It can be done at the same time as other screening tests, and on the same blood spot sample from the baby. This will allow babies with HCU to receive care and treatment much faster, before any symptoms occur.
“Our new test makes homocysteine analysis easier, solving a two-decades-old problem, and hopefully making cases like Elliott’s less common. The test increases the specificity for HCU screening,” explains Austin Pickens, PhD, research and development team lead for CDC’s Biochemical Mass Spectrometry Laboratory.
The CDC study on HCU screening was published in early 2023 in the American Association for Clinical Chemistry’s Clinical Chemistry journal. “This is a significant step to greatly improve quality of life for infants with HCU,” declares Konstantinos Petritis, PhD, chief of CDC’s Biochemical Mass Spectrometry Laboratory.
Accurate testing means that care and treatment can start immediately, allowing newborns to avoid lifelong health problems or an early death.
Early Diagnosis Is Key
The Division of Laboratory Sciences in CDC’s National Center for Environmental Health plays an important role in newborn screening.
CDC has the world’s only laboratory enhancing the quality and accuracy of newborn screening test results across the United States and more than 80 countries. Every state in the U.S. screens newborns for many serious but treatable congenital diseases. These range from spinal muscular atrophy, cystic fibrosis, and sickle cell disease to endocrine diseases and multiple inborn errors of metabolism, including lysosomal storage diseases. Early and accurate testing allows babies to be diagnosed and treated promptly.
‘Serious But Treatable’
“This new breakthrough is going to save lives,” declares Carter, who not only has a child with HCU, but also works as an advocate for newborn screening.
“Thanks to CDC researchers for the incredible work they are doing! We think half of all babies with HCU are missed. That’s huge. I’m hoping that number goes down to zero. It is a manageable disorder—as our doctor said, serious but treatable.”
She calls her son the new and improved “Elliott 2.0.” Now 7, Elliott is happy and thriving. He excels in first grade. He plays flag football and basketball. He does everything his big brother, Grayson, does—and wants to do it better. Grayson doesn’t have HCU, she adds.
As a very proud mom, Carter tears up when she says, “Elliott has beyond exceeded what I wondered and worried about in the hospital. Thank God. We were very lucky.”
Danae Bartke, executive director of HCU Network America as well as a patient herself, also takes CDC’s new breakthrough research personally. Her brother, Garrett Austin, 33, also has HCU. She has led HCU Network America’s charge for improvement to newborn screening since the organization’s founding in 2016.
“The news of a first-tier test is nothing short of a miracle in our eyes,” says Bartke. “We are incredibly grateful for CDC’s hard work, attention, and dedication to our community.”
She wants all families to have the best possible outcome, and says it starts with effective newborn screening.