COVID-19 Science Update released: December 3, 2021 Edition 115

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 database, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Section headings in the COVID-19 Science Update have been changed to align
with the CDC Science Agenda for COVID-19.
Here you can find all previous COVID-19 Science Updates.
PREPRINTS (NOT PEER-REVIEWED)
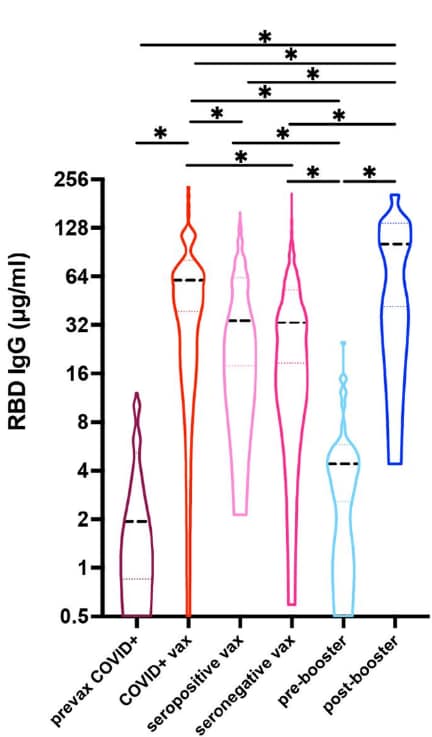
Antibody titers before and after booster doses of SARS-CoV-2 mRNA vaccines in healthy adults. Demonbreun et al. medRxiv (November 21, 2021).
Key findings:
- Adults who received a booster dose of mRNA COVID-19 vaccine 182–290 days after a primary series had a ~25-fold increase in anti-RBD IgG concentration relative to pre-booster (Figure).
- Median antibody concentration was significantly higher post-booster than after infection, after a primary 2-dose series among infection-naïve individuals, and after a primary series among previously infected individuals.
- Post-booster IgG levels were similar in females and males, and lower with older age.
- Post-booster neutralization titers were high for the SARS-CoV-2 Delta (B.1.617.2) variant and higher for wild-type SARS-CoV-2.
Methods: Fingerstick dried blood spot specimens were collected from adults previously fully vaccinated with an mRNA COVID-19 vaccine series (n = 33) before and 6–10 days after booster dose administration (mean 237.9 days after last dose). Antibody (anti-RBD IgG) and surrogate virus neutralization (spike-ACE2 receptor interaction) results were compared to results from a prior community-based study that used the same protocols to quantify antibody responses after diagnosis of COVID-19 or administration of COVID-19 vaccine. Limitations: Small sample size; lack of stratification by mRNA vaccine type and long-term immune response assessment.
Implications: In this study, mRNA COVID-19 vaccine boosters augmented immune responses to SARS-CoV-2 among individuals already vaccinated against and/or exposed to SARS-CoV-2. CDC now recommends that everyone ages ≥18 years should receive a COVID-19 vaccine booster.
Figure:
Note: Adapted from Demonbreun et al. Median antibody (anti-RBD IgG) concentrations among adults pre-booster or post-booster (n = 33) compared to those among adults with outpatient COVID-19 who were unvaccinated (prevax COVID+), adults with outpatient COVID-19 who were vaccinated (COVID+ vax), adults with unknown COVID-19 history who were seropositive and vaccinated (seropositive vax), and adults with unknown COVID-19 history who were seronegative and vaccinated (seronegative vax). Black dashed lines indicate median, colored dotted lines indicate 25th and 75th percentiles, *p <0.001. Licensed under CC-BY-NC-ND 4.0.
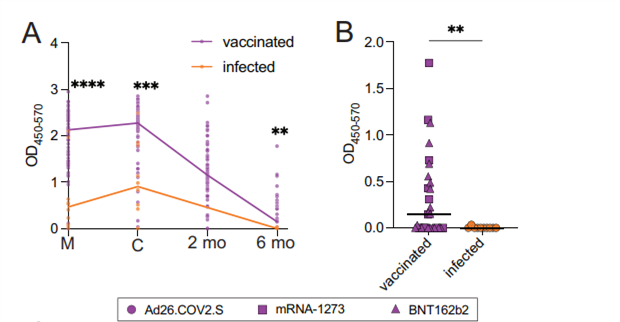
Durability of anti-spike antibodies in the infant after maternal COVID-19 vaccination. Shook et al. medRxiv (November 20, 2021). Published in JAMA as Durability of anti-spike antibodies in infants after maternal COVID-19 vaccination or natural infection (February 7, 2022).
Key findings:
- Among infants born to participants who received COVID-19 vaccination during pregnancy, 94% (58/62) had detectable antibodies (anti-spike IgG) at age 2 months and 60% (18/30) at age 6 months.
- In contrast, among infants born to participants who were infected with SARS-CoV-2 during pregnancy, only 8% (1/12) had antibodies at age 6 months.
- Among infants born to participants who were vaccinated during pregnancy, infant titers at age 2 months were correlated with titers from maternal and cord blood at delivery.
Methods: Cross-sectional study in Massachusetts that included 90 pregnant participants who received COVID-19 vaccination and 12 pregnant participants who were infected with SARS-CoV-2 during pregnancy. Matched maternal and umbilical cord sera were collected during the delivery hospitalization, and liveborn infants of enrolled pregnant participants were enrolled after birth. Specimen collection was in summer and early fall 2021. Limitations: Small sample size; race/ethnicity data unavailable; breastfeeding not assessed.
Implications: COVID-19 vaccination during pregnancy resulted in a higher proportion of infants with detectable antibodies for at least 6 months. Findings support COVID-19 vaccination recommendations for people who are pregnant and suggest that protection might extend to their unvaccinated infants.
Figure:
Note: Adapted from Shook et al. A) Antibody (anti-spike IgG) titers in maternal sera at delivery (M), umbilical cord sera at delivery (C), infant sera at age 2 months (2 mo), and infant sera at age 6 months (6 mo) in vaccinated mothers or unvaccinated mothers with SARS-CoV-2 infection during pregnancy. B) Antibody titers in infants at age 6 months with vaccinated mothers or unvaccinated infected mothers. Differences assessed with Mann-Whitney test, ** p<0.01, *** p<0.001, ****p<0.0001. Used by permission of authors.
PREPRINTS (NOT PEER-REVIEWED)
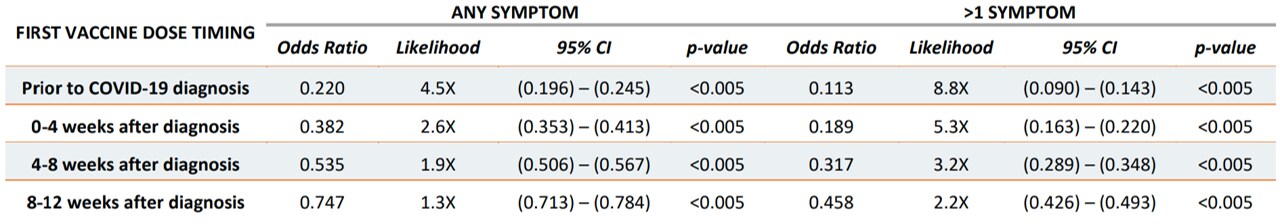
Reduced incidence of long-COVID symptoms related to administration of COVID-19 vaccines both before COVID-19 diagnosis and up to 12 weeks after. Simon et al. medRxiv (November 18, 2021).
Key findings:
- Compared with unvaccinated persons, persons vaccinated before and up to 12 weeks after a COVID-19 diagnosis had lower odds of experiencing ≥1 symptom of long COVID (Figure).
- Persons vaccinated before their COVID-19 diagnosis were the least likely to experience symptoms of long COVID (OR 0.220, 95% CI 0.196-0.245).
- Persons with shorter intervals to vaccination following their COVID-19 diagnosis had lower odds of experiencing long COVID symptoms compared with persons with longer intervals to vaccination.
Methods: A retrospective study of persons with COVID-19 (n = 240,648) during February 2020–May 2021 was conducted using electronic health records and claims data. Long COVID cases were defined as persons who received medical care for ≥1 COVID-associated symptom 12–20 weeks after the initial COVID-19 diagnosis. ORs were based on logistic regression and adjusted for demographics and underlying health conditions. Limitations: Conducted before the Delta (B.1.617.2) variant became predominant; does not evaluate vaccine product.
Implications: COVID-19 vaccination, when administered before or shortly after a COVID-19 diagnosis, might protect against symptoms of long COVID.
Figure:
Note: Adapted from Simon et al. Odds of long COVID symptoms were lower among vaccinated persons (stratified by timing of vaccination) relative to persons who remained unvaccinated up to 12 weeks after diagnosis. Likelihood values indicate reduction in likelihood of developing long COVID symptoms relative to unvaccinated persons. Licensed under CC-BY-NC-ND 4.0.
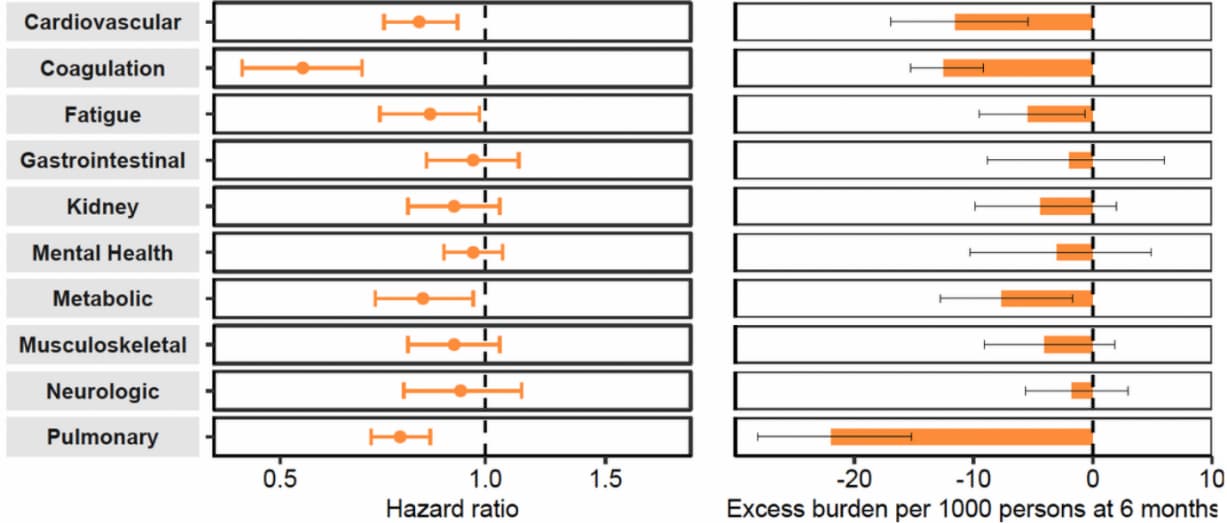
Long COVID after breakthrough COVID-19: The post-acute sequelae of breakthrough COVID-19. Al-Aly et al. Research Square (November 15, 2021). Published in Nature Medicine as Long COVID after breakthrough SARS-CoV-2 infection (May 25, 2022).
Key findings:
- Compared with unvaccinated persons with SARS-CoV-2 infection, persons with breakthrough infection had lower risk of death (HR 0.65, 95% CI 0.55-0.76) and lower risk of post-acute sequelae (HR 0.87, 95% CI 0.83-0.92) 30 days to 6 months after infection (Figure).
- Compared with persons who did not have SARS-CoV-2 infection, persons with breakthrough infection had higher risk of death (hazard ratio [HR] 1.53, 95% CI 1.36-1.72) and higher risk of ≥1 post-acute sequelae (HR 1.59, 95% CI 1.53-1.65) 30 days to 6 months after infection.
Methods: U.S. Department of Veterans Affairs electronic health records data were used to determine risk of death and risk of ≥1 post-acute sequela among persons with a documented first positive SARS-CoV-2 test result after COVID-19 vaccination (“breakthrough,” n = 16,035) compared with persons with a positive SARS-CoV-2 test result with no prior COVID-19 vaccination (n = 48,536), and a control group with no positive SARS-CoV-2 test result (n = 3,569,525) during February–October 2021. HRs were adjusted for demographics and health characteristics. Limitations: Veterans not representative of the general U.S. population; infection status of persons who were not tested might be misclassified.
Implications: People with SARS-CoV-2 infections appear to be at risk for long-term sequelae during the 6 months following infection. COVID-19 vaccination might reduce, but not eliminate, these risks.
Figure:
Note: Adapted from Al-Aly et al. Risk and 6-month excess burden of post-acute sequelae, by organ system, among people with breakthrough SARS-CoV-2 infection compared with unvaccinated infected persons. Adjusted hazard ratios (circles) and excess burden (bars) are displayed. Error bars indicate 95% CIs. Licensed under CC BY 4.0.
PREPRINTS (NOT PEER-REVIEWED)
Effectiveness of BNT162b2 and ChAdOx1 against SARS-CoV-2 household transmission: A prospective cohort study in England. Clifford et al. medRxiv (November 25, 2021).
Key findings:
- Among household contacts of adult (age ≥18 years) index cases with SARS-CoV-2 infection in the United Kingdom, estimated vaccine effectiveness (VE) of 2 doses of BNT162b2 (Comirnaty, Pfizer/BioNTech) was 71% (95% credible interval [CrI] 12%-95%) against Alpha (B.1.1.7) and 24% (95% CrI −2%-64%) against Delta (B.1.617.2) infection.
- Among adult index cases, estimated VE against onward transmission was 57% for Alpha and 31% for Delta infection.
Methods: During February–September 2021, adult index cases with SARS-CoV-2 infection and their household contacts were identified from community testing data; participants used self-testing kits to take oral-nasal swabs on days 1, 3, and 7 after enrollment that were tested by RT-qPCR; Bayesian logistic regression models were adjusted for age, vaccination history, and variant. Limitations: Limited power to detect differences in VE; did not assess or adjust for possible waning vaccine immunity relative to the circulation of the two variants; unable to assess VE among younger age groups (persons aged <18 years were not eligible for vaccination at the time).
Implications: COVID-19 vaccination was effective in reducing household transmission of SARS-CoV-2, although protection might be lower against Delta than Alpha variants.
Two studies evaluated COVID-19 mortality in the United States by race, ethnicity, and education.
PEER-REVIEWED
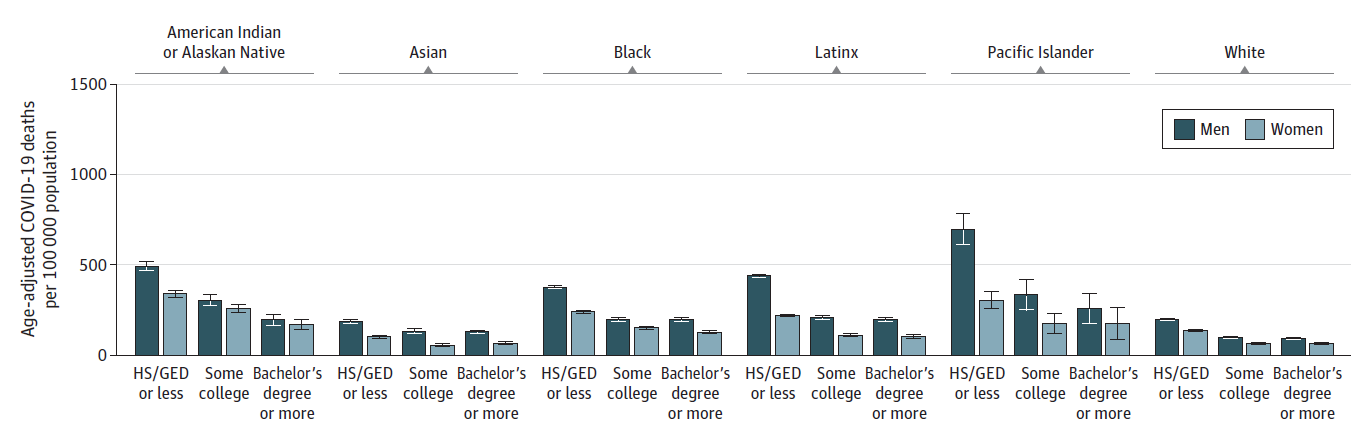
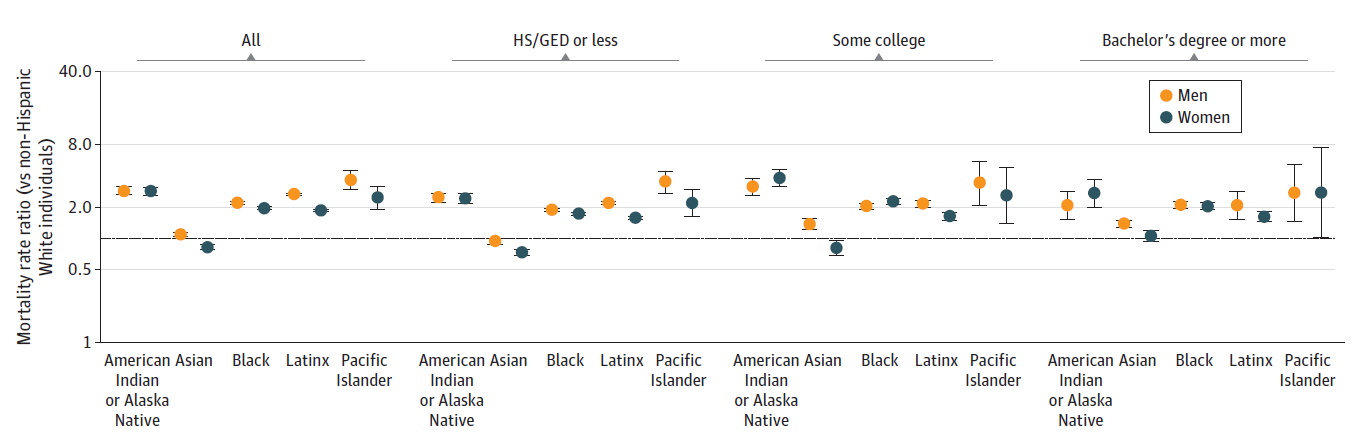
A. Variation in COVID-19 mortality in the US by race and ethnicity and educational attainment. Feldman et al. JAMA Network Open (November 23, 2021).
Key findings:
- Among persons aged ≥25 years in the United States, age-adjusted cumulative mortality rates from COVID-19 in 2020 varied by race/ethnicity and educational attainment, ranging from 54.4 per 100,000 among Asian women with some college education to 699.0 per 100,000 among Native Hawaiian and Other Pacific Islander men with a high school degree or less (Figure).
- Within each race and ethnicity group, mortality rates were highest among persons with lower educational attainment.
- When comparing within levels of education groups, most race and ethnicity minority groups had higher age-adjusted mortality rates than their non-Hispanic White counterparts.
Methods: Cross-sectional study determined COVID-19 cumulative mortality among all persons aged ≥25 years in the United States in 2020 (n = 219 million) using open access data provided by CDC. Mortality was determined in subgroups jointly stratified by age, sex, race and ethnicity, and educational attainment. Limitations: Data could be subject to misclassification of race, ethnicity, or cause of death; unable to assess potential modifiers (e.g., geography) or changes in mortality over time.
Figure:
A.
Note: Adapted from Feldman et al. COVID-19 (A) cumulative mortality rates by race and ethnicity, sex (men, women), and educational attainment and (B) mortality rate ratios by race and ethnicity, sex (men, women), and educational attainment, for all persons aged ≥25 years in the United States in 2020. HS/GED = high school or General Educational Development certification. Error bars indicate 95% CIs. In (B), comparisons are made to non-Hispanic White persons; black horizontal line indicates a mortality rate ratio of 1.0. Licensed under CC BY.
PREPRINTS (NOT PEER-REVIEWED)
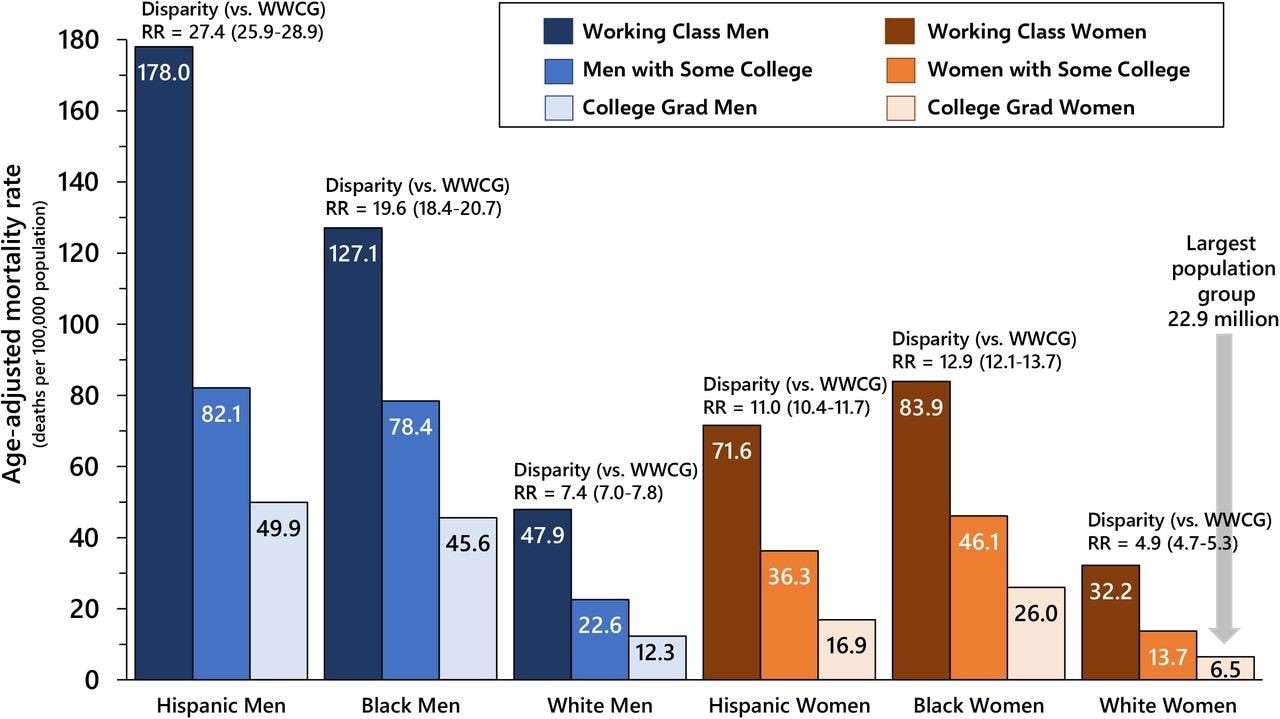
B. Social class, race/ethnicity, and COVID-19 mortality among working age adults in the United States. Pathak et al. medRxiv (November 24, 2021). Published in International Journal of Environmental Research and Public Health as Joint effects of socioeconomic position, race/ethnicity, and gender on COVID-19 mortality among working-age adults in the United States (April 30, 2022).
Key findings:
- During 2020, working-class adults (adults who did not receive any college education) were 3–5 times more likely to die from COVID-19 than their college-educated counterparts.
- Working-class Hispanic or Latino (RR 27.4, 95% CI 25.9-28.9) and Black or African American (RR 19.6, 95% CI 18.4-20.7) men had some of the highest risks for death from COVID-19 compared with college-educated White women.
- Working-class Black or African American women had nearly 13 times higher risk of death from COVID-19 (RR: 12.9, 95% CI 12.1-13.7) than college-educated White women.
- Mortality from COVID-19 was consistently higher among men (12.3–178.0 deaths per 100,000) than women (6.5–83.9 deaths per 100,000).
Methods: U.S. age-adjusted COVID-19 mortality rate study among adults aged 25–64 years by social class, race and ethnicity, and gender during 2020 using the National Center for Health Statistics (NCHS) ad hoc death certificate tabulation in February 2021. Educational attainment was used as a surrogate marker for social class. Limitations: Death certificate data might underreport COVID-19 mortality, with variation in accurate reporting over time.
Figure:
Note: Adapted from Pathak et al. COVID-19 mortality rates and social class-gender-race/ethnicity disparity rate ratios among Hispanic or Latino, Black or African American, and White men and women aged 25–64 years, United States, 2020. White women college graduates (WWCG) are the referent group for rate ratios. Used by permission of authors.
Implications for Feldman et al. and Pathak et al.: Public health interventions that reach and are adoptable by racial and ethnic minority populations and persons with less educational attainment are critical to minimizing mortality during the COVID-19 pandemic.
Variants
- SARS-CoV-2 sequencing update. Network for Genomic Surveillance South Africa (NGS-SA). Media presentation (November 25, 2021). South Africa released initial data on a new SARS-CoV-2 variant (lineage B.1.1.529) with a number of mutations. The World Health Organization (WHO) and the U.S. government SARS-CoV-2 Interagency Group (SIG) subsequently designated B.1.1.529 a variant of concern, named Omicron.
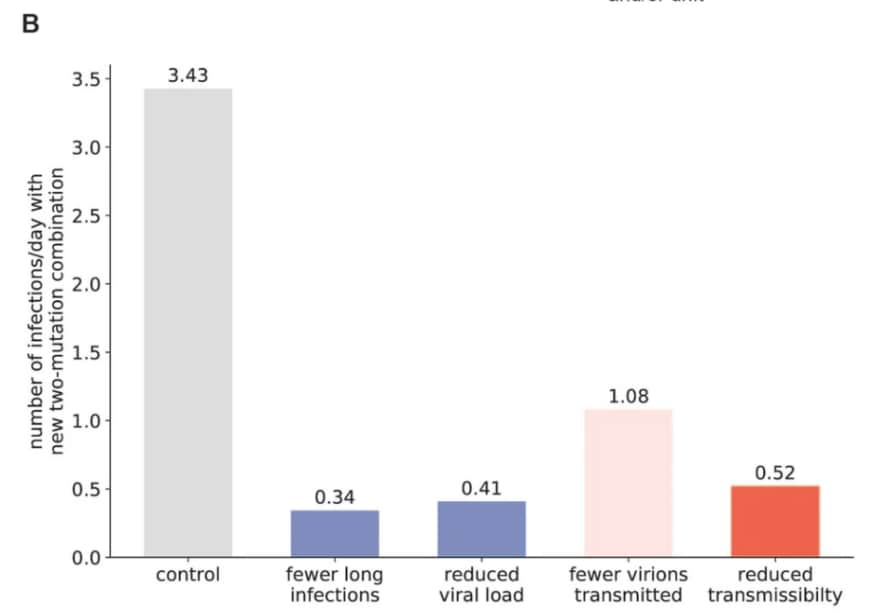
- Controlling long‑term SARS‑CoV‑2 infections can slow viral evolution and reduce the risk of treatment failure. Van Egeren et al. Scientific Reports (November 19, 2021). An evolutionary model found that higher SARS-CoV-2 viral load and longer duration of infection favor emergence of fitter SARS-CoV-2 variants within an infected person. Control strategies that reduce SARS-CoV-2 viral load and minimize transmission to and from persons at increased risk of long-term infections (for example, immunocompromised persons) could slow the emergence and spread of new variants.
Note: Adapted from Van Egeren et al. A) Schematic showing events necessary for generation and population-level establishment of a new SARS-CoV-2 variant. B) Results of modeling efforts looking at the reduction in generation rate of hypothetical new double mutant lineages after various interventions to reduce transmission and slow viral evolution. Licensed under CC BY 4.0.
Vaccines
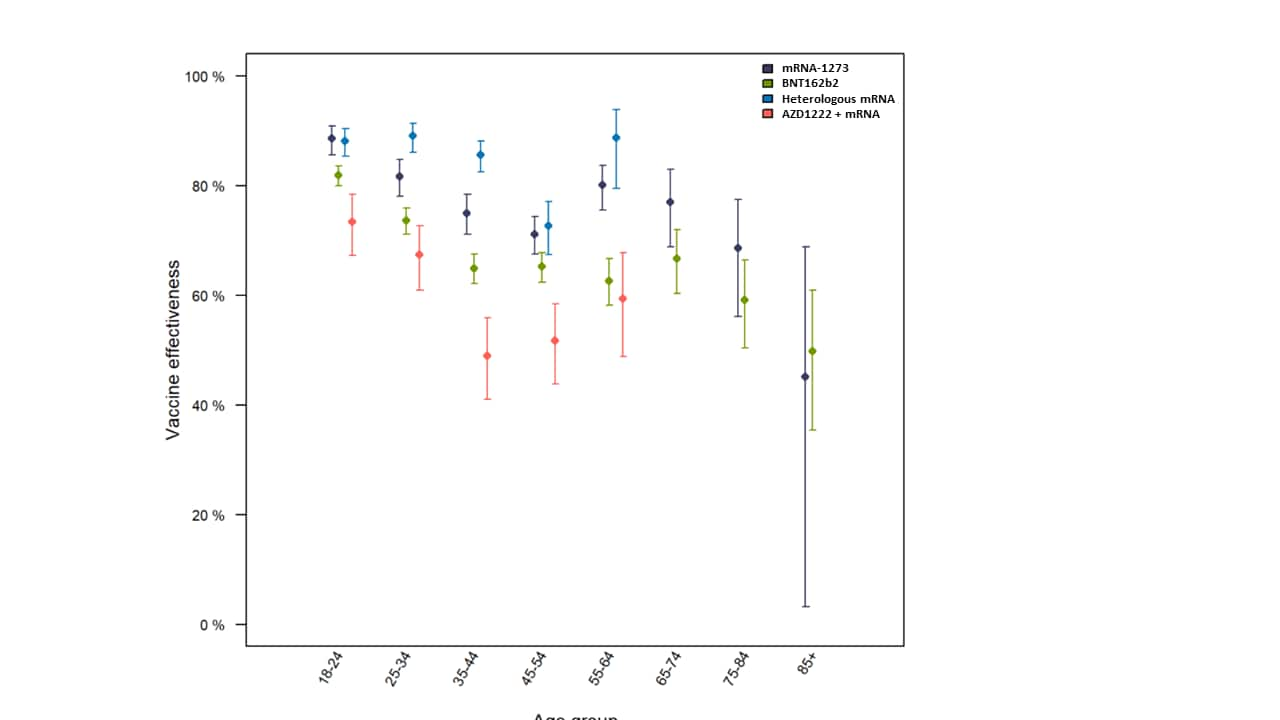
- Age and product dependent vaccine effectiveness against SARS-CoV-2 infection and hospitalisation among adults in Norway: A national cohort study, January – September 2021. Starrfelt et al. medRxiv (Preprint; November 12, 2021). Among 4.3 million adults in Norway during January–September 2021, adjusted vaccine effectiveness (VE) against infection among fully vaccinated persons was highest among those who received a heterologous mRNA COVID-19 vaccine series (VE 84.7%, 95% CI 83.1%-86.1%), followed by mRNA-1273 (VE 78.2%, 95% CI 76.7%-79.6%), and BNT162b2 (VE 69.7%, 95% CI 68.6%-70.8%) vaccine series.
Note: Adapted from Starrfelt et al. Adjusted vaccine effectiveness against SARS-CoV-2 infection by age group and vaccine (mRNA-1273, BNT162b2, heterologous mRNA, and AZD1222 + mRNA). Adjusted for sex, underlying medical conditions, county of residence, country of birth, and crowding. Licensed under CC-BY-ND 4.0.
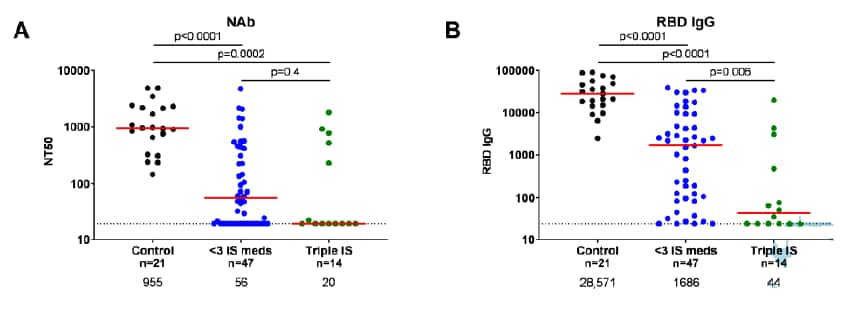
- COVID-19 mRNA vaccine immunogenicity in immunosuppressed individuals. Collier et al. Journal of Infectious Diseases (November 18, 2021). Following mRNA COVID-19 vaccination, persons receiving immunosuppressive therapy had lower neutralizing, binding, and non-neutralizing antibody functions than immunocompetent persons. Immunosuppressive therapy was also associated with lower CD4 and CD8 T cell IFN-γ responses and lower cross-reactivity against SARS-CoV-2 variants.
Note: Adapted from Collier et al. A) Pseudovirus neutralizing antibody titers (NT50) and B) SARS-CoV-2 spike receptor binding domain (RBD) IgG titers 2–8 weeks following 2nd dose of mRNA COVID-19 vaccination in persons receiving single/double immunosuppression (IS) therapy, persons receiving triple IS therapy, and controls. Licensed under CC-BY-NC-ND.
Natural History, Reinfection, and Health Impact
- Severity of SARS-CoV-2 reinfections as compared with primary infections. Abu-Raddad et al. NEJM (November 24, 2021). Among unvaccinated persons with PCR-confirmed SARS-CoV-2 infection in Qatar during February 2020–April 2021, reinfections were associated with lower odds of severe disease (OR 0.12, 95% CI 0.03-0.31) than initial infections. Among persons with reinfection, odds of a composite outcome of severe, critical, and/or fatal disease were 0.10 times (95% CI 0.03-0.25) that among persons with primary infection. No cases of critical or fatal disease were identified among 1,300 persons with re-infections.
- An immune correlate of SARS-CoV-2 infection and severity of reinfections. Maier et al. medRxiv (Preprint; November 24, 2021). In a household cohort study in Nicaragua (n = 2,123), previous SARS-CoV-2 infection was associated with 64.5% protection from re-infection during March–October 2021 (95% CI 56.4%-71.1%), 69.2% protection from symptomatic infection (95% CI 60.7%-75.9%), and 79.4% protection from moderate or severe infection (95% CI 64.9%-87.9%). Compared with first infections (n = 377), re-infections (n = 162) were less likely to be associated with moderate or severe disease (RR 0.6, 95% CI 0.38-0.98) and more likely to be associated with subclinical disease (RR 1.9, 95% CI 1.33-2.73).
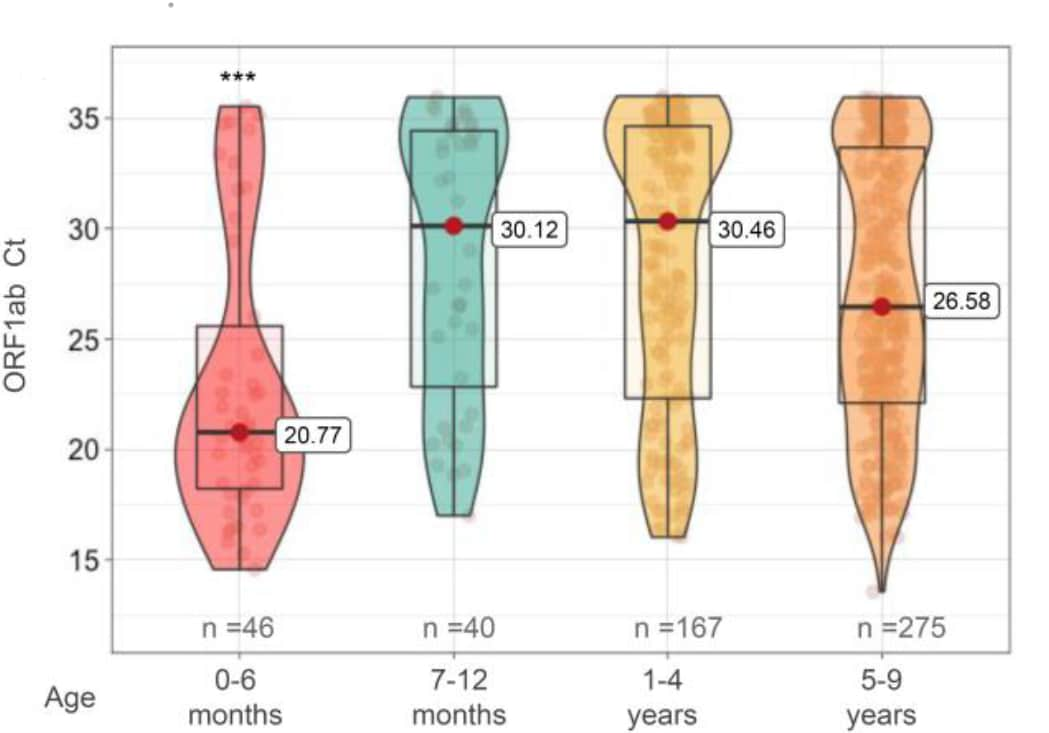
- Infants younger than 6 months old infected by SARS-CoV-2 show the highest respiratory viral loads. Ochoa et al. Journal of Infectious Diseases (November 24, 2021). Among 45,318 SARS-CoV-2-positive nasopharyngeal swab specimens obtained in Argentina, higher SARS-CoV-2 viral loads were suggested by lower Ct values. Infants age <6 months had the lowest median Ct (20.77, IQR 18.1-26.87) compared with any other age group, including adults (p <0.001). Median Ct was 6–10 cycles lower among infants age <6 months than among infants ages 7–12 months (30.12, IQR 22.01-34.56), children ages 1–4 years (30.46, IQR 22.34-34.69), or children ages 5–9 years (26.58, IQR 22.34-33.8).
Note: Adapted from Ochoa et al. SARS-CoV-2 Ct values were lower (suggesting higher viral loads) among infants ages <6 months, compared with infants ages 7–12 months, children ages 1–4 years, and children ages 5-9 years. *** p <0.001 for ages 0-6 months vs. any other age group. Used by permission of Oxford University Press for the Infectious Diseases Society of America.
From the Morbidity and Mortality Weekly Report (November 26, 2021).
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.