COVID-19 Science Update released: August 4, 2020 Edition 36

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Here you can find all previous COVID-19 Science Updates.
PEER-REVIEWED
A large COVID-19 outbreak in a high school 10 days after schools’ reopening, Israel, May 2020external icon. Stein-Zamir et al. Eurosurveillance (July 21, 2020).
Key findings:
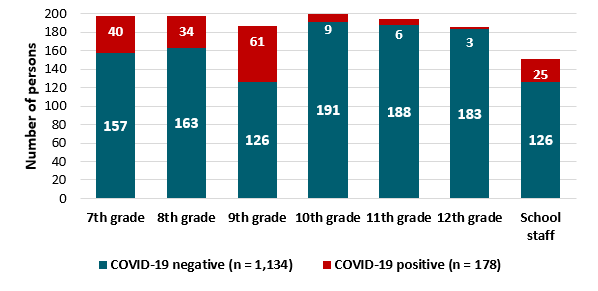
- 153 students (attack rate: 13.2%) and 25 staff members (attack rate: 16.6%) tested positive for SARS-CoV-2 (Figure).
- Highest proportion of cases were among 9th graders (32.6%).
- Symptoms were reported in 43% of students and 76% of staff, with no hospitalizations.
- Crowded class size and exemption of facemask wearing due to extreme heat are factors that probably contributed to transmission.
Methods: Outbreak investigation in an Israeli school after 2 COVID-19 cases were reported May 26-27, 2020. From May 28-30, school-wide COVID-19 testing was conducted among 151 staff and 1,161 students. An environmental school inspection was conducted to assess contributors to the outbreak. Limitations: Findings are not representative of other school outbreaks. There were limited data about contacts of COVID-19 cases outside of the school.
Implications: School reopening may result in COVID-19 outbreaks. Strict measures, particularly reducing class size and requiring students to wear facemasks, should be put in place when considering opening schools.
Figure:
Note: Adapted from Stein-Zamir et al. Results of COVID-19 testing, school outbreak, (n = 1,312) by grade, with negative laboratory results in dark teal and positive laboratory results in red. Licensed under CC-BY 4.0.
PREPRINTS (NOT PEER-REVIEWED)
Evaluating scenarios for school reopening under COVID19external icon. Keskinocak et al. medRxiv (July 24, 2020).
Key findings:
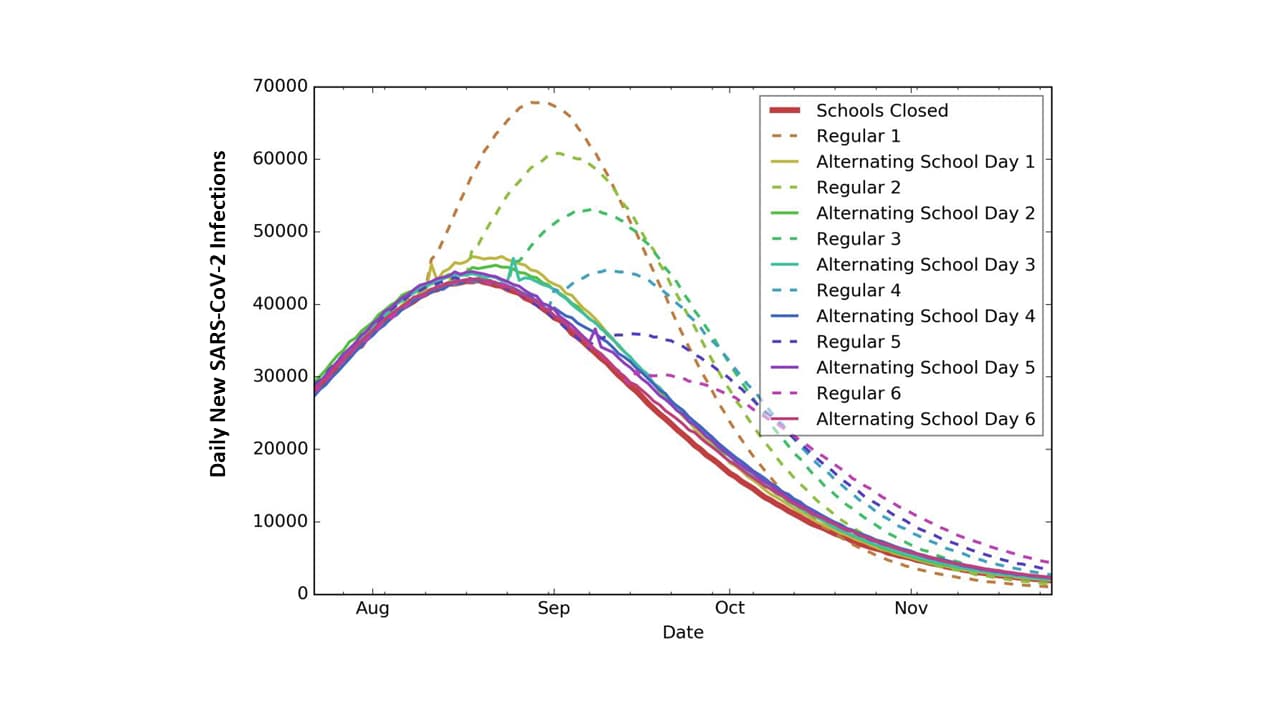
- Across all simulated school opening scenarios in the state of Georgia, the peak number of new infections was estimated to range from 43,000 to 68,000 during the study period (Figure).
- Under a regular school schedule, delaying the reopening date could delay the peak day and reduce the peak number of new infections.
- Delaying reopening would have minimal impact on alternating day schedules.
- An alternating school day schedule, which produced an infection attack rate (IAR) ranging from 38.5% to 39.8% for different opening dates, performed almost as well as keeping schools closed (IAR: 37.7%) and better than opening schools with a regular schedule (IAR range by opening dates: 42.4%-47.2%).
Methods: An agent-based simulation modelexternal icon was used to project the number of infections and deaths under multiple school reopening dates and scenarios. The differing cohorts assessed included: in-person instruction on alternating days, only younger children returning to in-person instruction, regular schedule for all students, and school closure (only online instruction). The simulated study period was from February 18, which was considered the start of voluntary quarantine, until November 24, 2020 and used the state of Georgia as the case study. Limitations: Did not consider the use of face masks or testing and contact tracing.
Implications: Reopening schools without strict public health measures may significantly increase the number of infections and deaths; alternating school day schedules may reduce transmission.
Figure:
Note: Adapted from Keskinocak et al. Comparison of daily number of new SARS-CoV-2 infections under different school reopening scenarios: schools closed (red line), schools opened following a regular schedule (dotted lines), and schools opened with an alternating school day schedule (straight lines). Numbers 1-6 refer to reopening schools on August 10, August 17, August 24, August 31, September 7, and September 14, respectively. Licensed under CC-BY-NC-ND 4.0.
PEER-REVIEWED
Systematic SARS-CoV-2 screening at hospital admission in children: A French prospective multicenter studyexternal icon. Poline et al. Clinical Infectious Disease (July 25, 2020).
Key findings:
- A symptom-based testing strategy for COVID-19 failed to detect 45% of infected children.
- 22/438 (5%) hospitalized pediatric patients were SARS-CoV-2-positive.
- 10/22 (45%) were asymptomatic.
- Screening children to detect infections with a comprehensive set of signs and symptoms performed poorly: sensitivity 55% (95% CI 32-76) and specificity 59% (95% CI 54-64).
- Screening children for a single clinical sign/symptom to detect COVID-19 also performed poorly with the following sensitivities: fever (36% [95% CI 17-59]), dyspnea (32% [95% CI 14-55]), skin involvement (23% [95% CI 8-45]), upper respiratory tract symptoms (32% [95% CI 14-55]), diarrhea/vomiting (41% [95% CI 21-64]).
Methods: Investigators tested 438 patients for SARS-CoV-2 infection by RT-PCR in four pediatric hospitals in France, April 15-30, 2020 before admission to determine the proportion of asymptomatically infected children. They also assessed the performance of a symptom-based screening strategy to identify children with COVID-19. Limitations: No follow-up testing to rule out false negative results; ages of children not reported.
Implications: Symptom-based screening of hospitalized pediatric patients for COVID-19 has limited utility. Systematic testing of all admitted children is recommended to limit nosocomial transmission.
Well-being of parents and children during the COVID-19 pandemic: A national surveyexternal icon. Patrick et al. Pediatrics. (July 1, 2020).
Key findings:
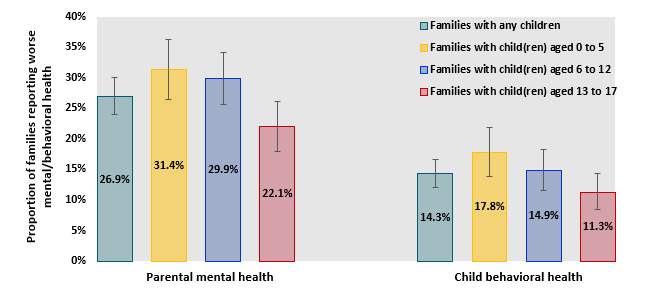
- In a survey of US parents, 9% reported worsening of their mental health since the pandemic began and 14.3% of reported worsening of their children’s behavioral health.
- Families with younger children were more likely to report worse parental mental health and worse child behavioral health (Figure 1).
- Female were more likely than male parents to report worsened mental health but declines were similar by race/ethnicity, income, educational achievement, and US Census regions.
- Physical health worsened for 17.7% (95% CI 15.0-20.3%) of parents and 3.8% (95% CI 2.5-5.1%) of children.
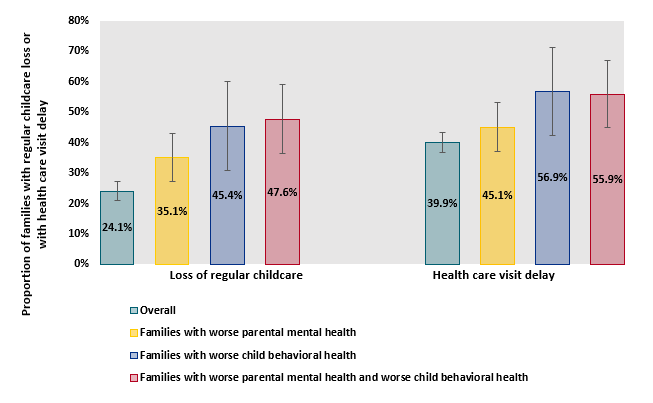
- 1% (95% CI 21.1-27.1%) of parents lost regular childcare and 39.9% (95% CI 36.6-43.2%) experienced health care visit delays during the pandemic.
- Among families reporting both worsening parental mental health and child behavioral health, 47.6% (95% CI 36.3-58.9%) lost regular childcare during the pandemic (Figure 2).
Methods: National cross-sectional survey of parents in 1,011 US households with at least 1 child under age 18, June 5-10, 2020. Analyses were weighted to provide national estimates and adjusted to account for non-response to the survey. Limitations: Recall bias; self-reported health status; no longitudinal data.
Implications: Policies that support families with children, including childcare and behavioral health support, are needed to help mitigate effects of the pandemic on mental and physical health.
Figure 1
Note: Adapted from Patrick et al. Proportion of families with worse parental mental health and child behavioral health since March 2020, by age(s) of child(ren) in family; families with any children, child(ren) aged 0 to 5, child(ren) aged 6 to 12, child(ren) aged 13 to 17. Whisker bars show 95% CIs.
Figure 2
Note: Adapted from Patrick et al. Loss of childcare and health care delays after March 2020 among families overall, with worse parental mental health, worse child behavioral health, or both. Whisker bars show 95% CIs.
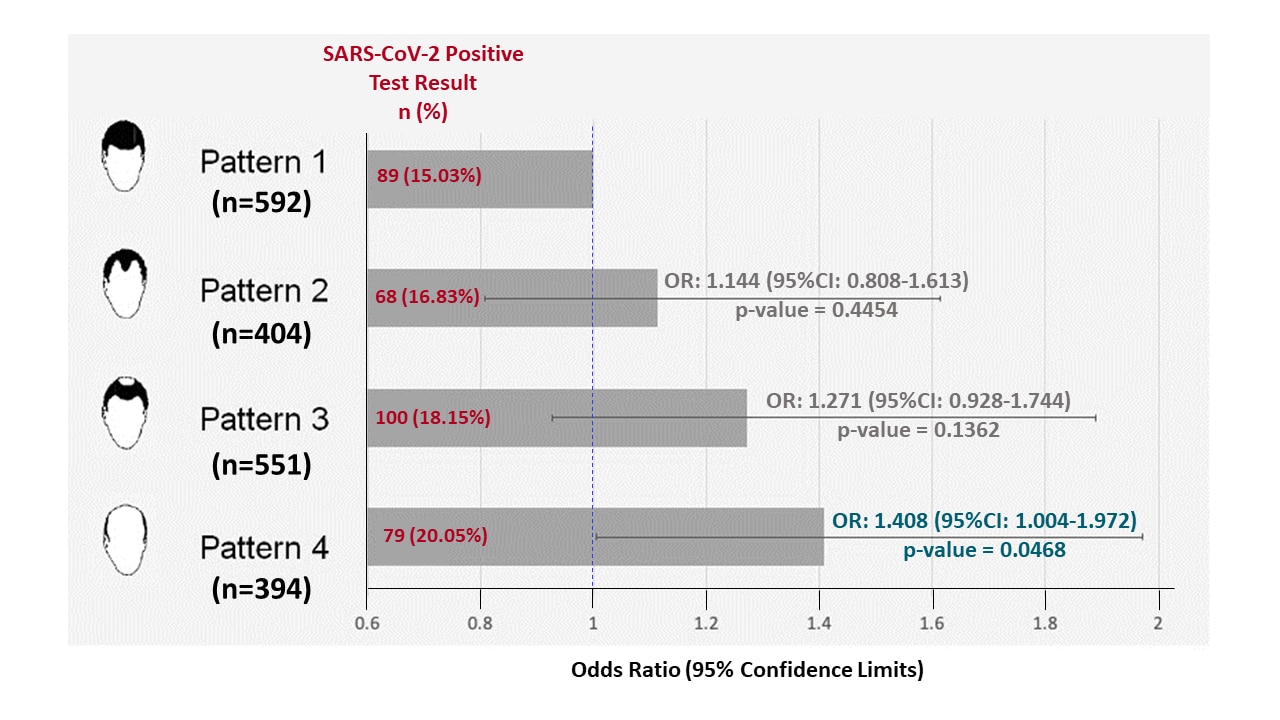
Male balding is a major risk factor for severe COVID-19external icon. Lee et al. Journal of the American Academy of Dermatology (July 22, 2020).
Key findings:
- Increasing extent of androgenic alopecia (balding) was associated with increased likelihood hospitalization for COVID-19 (Figure).
- Mean age and body mass index (BMI) were similar between those with and without COVID-19
Methods: 1,941 hospitalized men from the UK Biobank studyexternal icon, which collects biological samples and health data from 500, 000 persons in the UK, of whom 336 had COVID-19. Data from 2019 were used for all covariates (age, BMI, hypertension, dyslipidemia, diabetes, balding). Multivariable logistic regression was conducted to examine the association of balding with a positive SARS-CoV-2 test. Limitations: Balding data were self-reportedexternal icon; asymptomatic COVID-19 patients not included; no information provided on when participants were tested for SARS-CoV-2 or what test was used.
Implications: Severe androgenic alopecia might be associated with risk for more severeCOVID-19; more robust studies are needed.
Figure:
Note: Adapted from Lee et al. Odds ratios and 95% confidence intervals for balding patterns and covariates compared to Pattern 1 from a multivariable logistic regression analysis where the outcome is a positive SARS-CoV-2 test.
PEER-REVIEWED
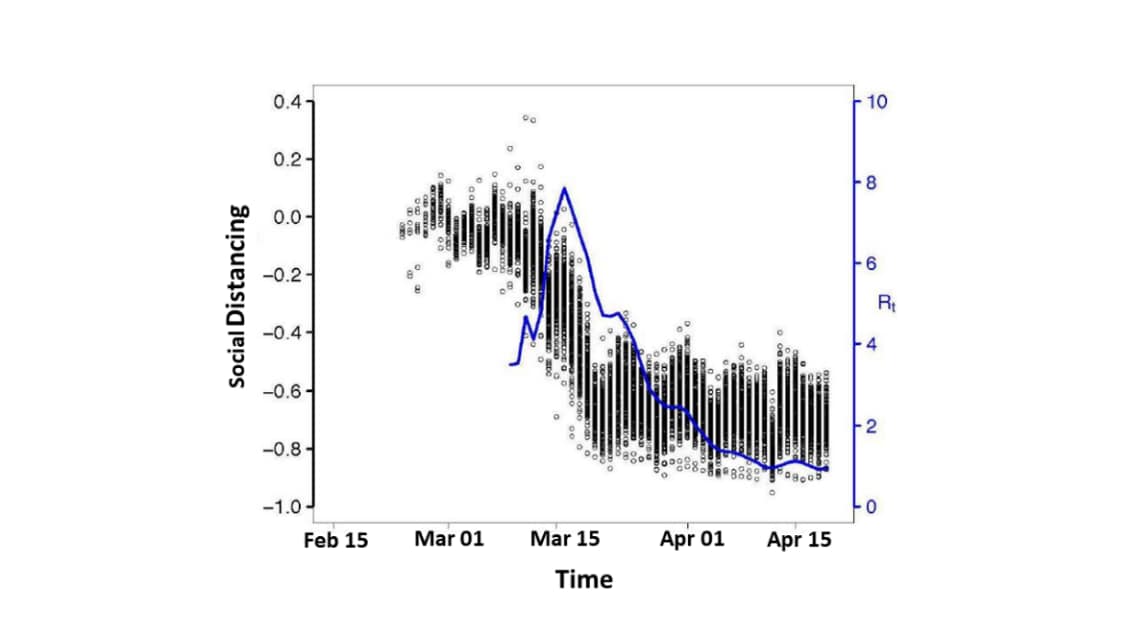
Association of social distancing, population density, and temperature with the instantaneous reproduction number of SARS-CoV-2 in counties across the United Statesexternal icon. Rubin et al. JAMA Network Open (July 23, 2020).
Key findings:
- The estimated instantaneous reproduction number (Rt) for SARS-CoV-2 varied across 211 US counties, reaching a peak of Rt of 7.8 in March and declining to approximately 1 in April (Figure 1).
- Social distancing was associated with the most substantial relative changes in Rt.
- 50% decrease in visits to nonessential businesses was associated with a 45% (95% CI, 43%-49%), decrease in Rt; at a 70% reduction in visits to nonessential business, 96% of counties had Rt < 1.0.
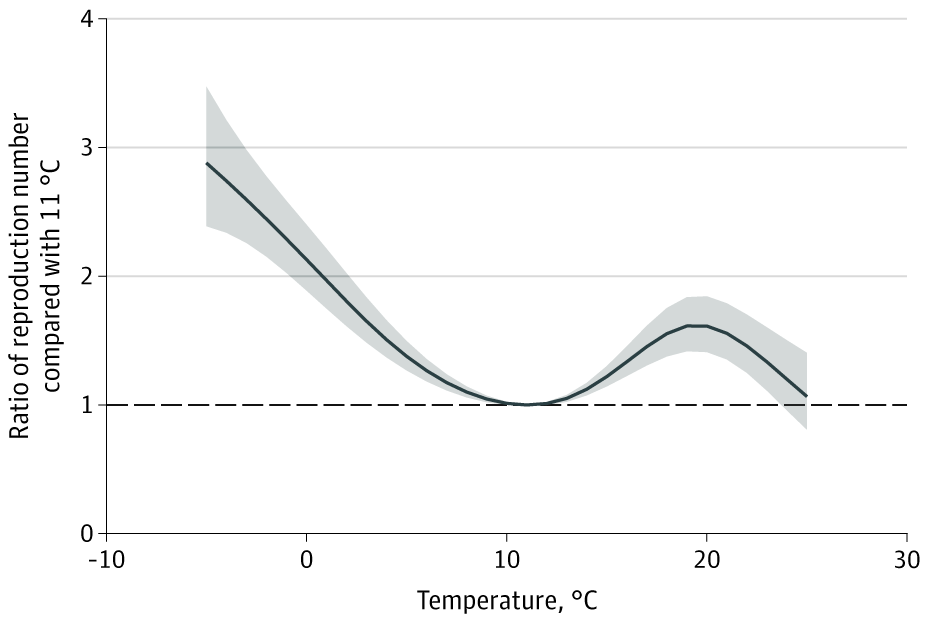
- Population density and wet-bulb daily temperatures were also associated with changes in Rt.
- 21 counties in the top decile of population density had a 15% increase (95% CI, 9%-22%; P < 0.001) in relative Rt compared with counties in the bottom quartile of population density.
- Wet-bulb temperature of 52°F (11°C) was associated with the lowest estimated Rt. Rt increased with peaks to 2.13 (95% CI, 1.89-2.40) at 32°F (0°C) and to 1.61 (95% CI, 1.41-1.84) at 68°F (20°C) (Figure 2).
Methods: Cohort study representing 54.8% of the US population that included 211 counties with >100,000 residents and at least 1 COVID-19 case, February 25-April 23, 2020. A hierarchical linear mixed-effects model was used to evaluate the association of percent change in visits to nonessential businesses measured by cellular telephone movement data, population density (obtained from US Census data) and daily wet-bulb temperature (a metric that captures both temperature and humidity) with Rt. Limitations: Limited generalizability to smaller counties as focus was on larger counties; mask use, gathering size, and long distance and international travel were not measured; temperature and humidity variations may be confounded by time period.
Implications: Social distancing was associated with substantial reductions in Rt; nearly all counties that achieved a 70% reduction in visits to non-essential businesses brought Rt below 1; lower population density, and potentially temperate weather appear to be associated with modestly reduced Rt.
Figure 1
Note: Adapted from Rubin et al. Rt of SARS CoV-2 (blue line; right y-axis) superimposed on scatter plot of reduction in visits to nonessential businesses (black circles; left y-axis) across 211 US counties over time (x-axis). Licensed under CC-BY.
Figure 2
Note: Adapted from Rubin et al. Cumulative exposure-response association between mean daily wet-bulb temperatures and Rt using a lag period of 4-14 days before case identification. The line represents the estimated Rt at each point along the temperature range compared with 11°C. The shaded areas represent the 95% CIs. The wet-bulb temperature range for the counties included in the analysis was −9 °C to 25°C. Licensed under CC-BY.
PEER-REVIEWED
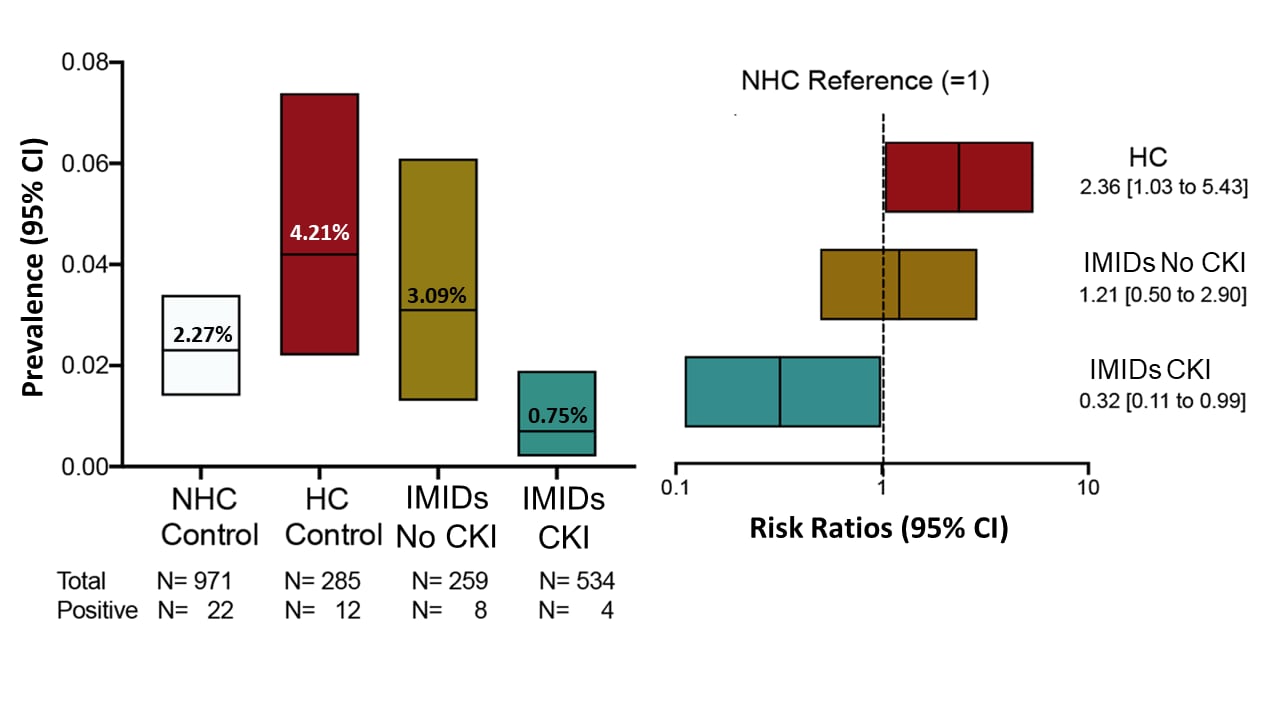
Patients with immune-mediated inflammatory diseases receiving cytokine inhibitors have low prevalence of SARS-CoV-2 seroconversionexternal icon. Simon et al. Nature Communications (July 24, 2020).
Key findings:
- The prevalence of anti-SARS-CoV-2 IgG antibodies in patients with immune-mediated inflammatory diseases (IMID) treated with cytokine inhibitors (0.7%) was lower than non-healthcare worker controls (NHCs, i.e., healthy persons without known IMID) (2.3%), (relative risk [RR] 0.32, 95% CI 0.11–0.99).
- The prevalence among IMID patients not treated with cytokine inhibitors (3.1%) did not differ significantly (RR 1.21, 95% CI 0.50-2.90) from prevalence in NHCs.
- The prevalence among health care professionals involved in the treatment of IMID patients (HC control) was significantly higher than among the NHC control cohort, indicating that their exposure to SARS-CoV-2 may be higher than in the general population (RR 2.36, 95% CI 1.03–5.43; p = 0.043)
Methods: Descriptive study of 753 patients with IMID (534 were on cytokine inhibitors for treatment of IMID, 259 were not treated with cytokine inhibitors for IMID), 285 health care professionals and 971 NHCs from multiple clinics in Germany (February-April 2020). Prevalence of IgG against the spike protein domain S1 was used to compare differences in infection rates among groups. Limitations: Low number of persons with positive serology; potential false positive antibody results; potential unmeasured confounders.
Implications: Cytokine inhibitor use in IMID patients may provide protection from SARS-CoV-2 infection; however, larger clinical studies are needed to confirm these results.
Figure:
Note: Adapted from Simon et al. Prevalence of anti-SARS-CoV-2 IgG antibodies across study groups. Left: Prevalence and 95% CIs of a positive anti-SARS-CoV-2 IgG antibody test recognizing the S1 domain of the spike protein in the non-health care (NHC) control cohort, health care (HC) control cohort and patients with immune-mediated inflammatory diseases not on cytokine inhibitor treatment (IMIDs No CKI) and those on treatment with cytokine inhibitors (IMIDs CKI). Right: Risk ratios and 95% CIs of anti-SARS-CoV-2 IgG antibody positivity in the HC control cohort, IMIDs No CKI, and IMIDs CKI with the NHC control cohort as reference. Licensed under CC-BY.
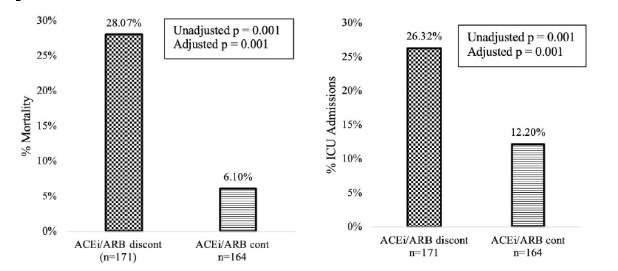
Continued in-hospital ACE inhibitor and ARB use in hypertensive COVID-19 patients is associated with positive clinical outcomesexternal icon. Lam et al. Journal of Infectious Diseases (July 23, 2020).
Key findings:
- In hypertensive patients hospitalized with COVID-19, in-hospital continuation of angiotensin-converting enzyme inhibitors (ACEi) or angiotensin II receptor blockers (ARBs) was associated with lower rates of mortality (28.1% vs 6.1%) and intensive care unit (ICU) admission (26.3% vs 12.2%) (Figure 1).
- Differences in mortality remained when patients who developed hypotension or acute kidney injury were excluded.
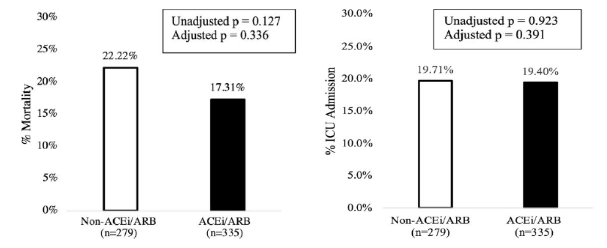
- Patients who were taking ACE inhibitors or ARBs at home did not have significantly different rates of mortality (22.2% vs 17.3%) or ICU admission (19.7% vs 19.4%) than patients not taking these medications at home (Figure 2).
- Compared with patients taking ACEi/ARBs at home, patients not taking ACEi/IRBs at home were older (73 vs 68 years) and less likely to have diabetes (35.1% vs 45.4%) but more likely to have chronic kidney disease (23.4% vs 9.0%).
Methods: Retrospective single-center study of 614 hospitalized COVID-19 patients with a history of hypertension, February 7-May 23, 2020. Patients were divided into 3 groups: A) patient not taking ACEi/ARBs at home B) patients taking ACEi/ARBs at home but had ACEi/ARBs discontinued during hospitalization C) patients who continued ACEi/ARBs during hospitalization. Age, gender, and comorbidities were included as covariates in logistic regression modeling. Limitations: Patients were not randomized; unmeasured confounders might have been associated with both discontinuation of ACEi/ARBs during hospitalization and adverse outcomes.
Implications: Although there have been concerns raised with using ACEi/ARBs in patients with COVID-19, this study provides evidence that stopping ACEi/ARBs in hypertensive patients to prevent adverse outcomes from COVID-19 is unwarranted.
Figure 1
Note: Adapted from Lam et al. Mortality (left) and ICU admission (right) rates in patients taking ACE inhibitors or ARBs at home and did (checkered bars) or did not (striped bars) have their medication discontinued in the hospital. Available via Oxford University Press Public Health Emergency Collection through PubMed Central.
Figure 2
Note: Adapted from Lam et al. Mortality (left) and ICU admission (right) rates in patients who were not (white bars) or were (black bars) taking ACE inhibitors or ARBs at home. Available via Oxford University Press Public Health Emergency Collection through PubMed Central.
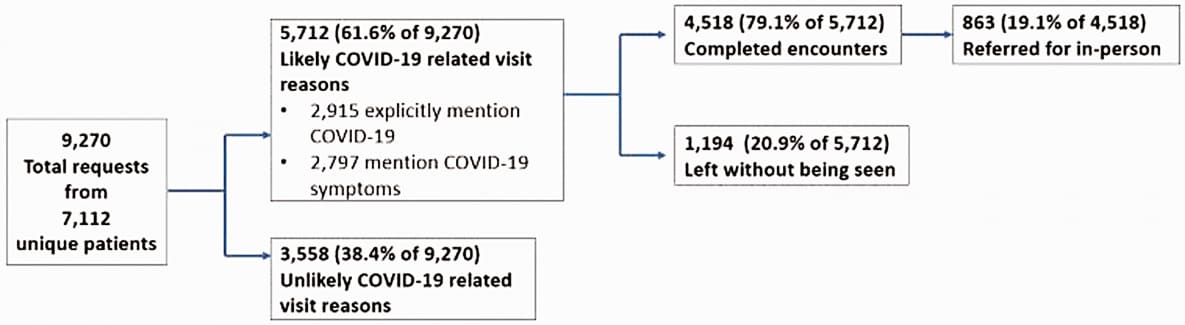
A descriptive analysis of an on-demand telehealth approach for remote COVID-19 patient screeningexternal icon. Ratwani et al. Journal of Telemedicine and Telecare (July 23, 2020).
Key findings:
- 5,712 (61.6%) of 9,270 on-demand telehealth requests were likely related to COVID-19 (Figure).
- Among the 4,518 completed on-demand telehealth encounters, 863 (19.1%) were referred for in-person services.
- The average completed encounter wait time was 26.5 minutes and the mean visit length was 8.8 minutes.
- Without on-demand telehealth, 42.8% of surveyed COVID-19 patients would have sought in-person care.
Methods: Descriptive analysis of 9,270 on-demand telehealth requests by 7,112 unique patients, between March 13 and April 3, 2020. A subset of patients with completed telehealth experience were surveyed on alternative options for healthcare. Limitations: Race and insurance information not available; referred patients were not followed up.
Implications: Telehealth services can provide care to patients with possible SARS-CoV-2 infection, limiting exposure to health care personnel and reducing the burden on the healthcare system.
Figure:
Note: Adapted from Ratwani et al. Flow diagram of likely COVID-19 patient encounters using on-demand telehealth. Used by permission of publisher via RightsLink.
PREPRINTS (NOT PEER-REVIEWED)
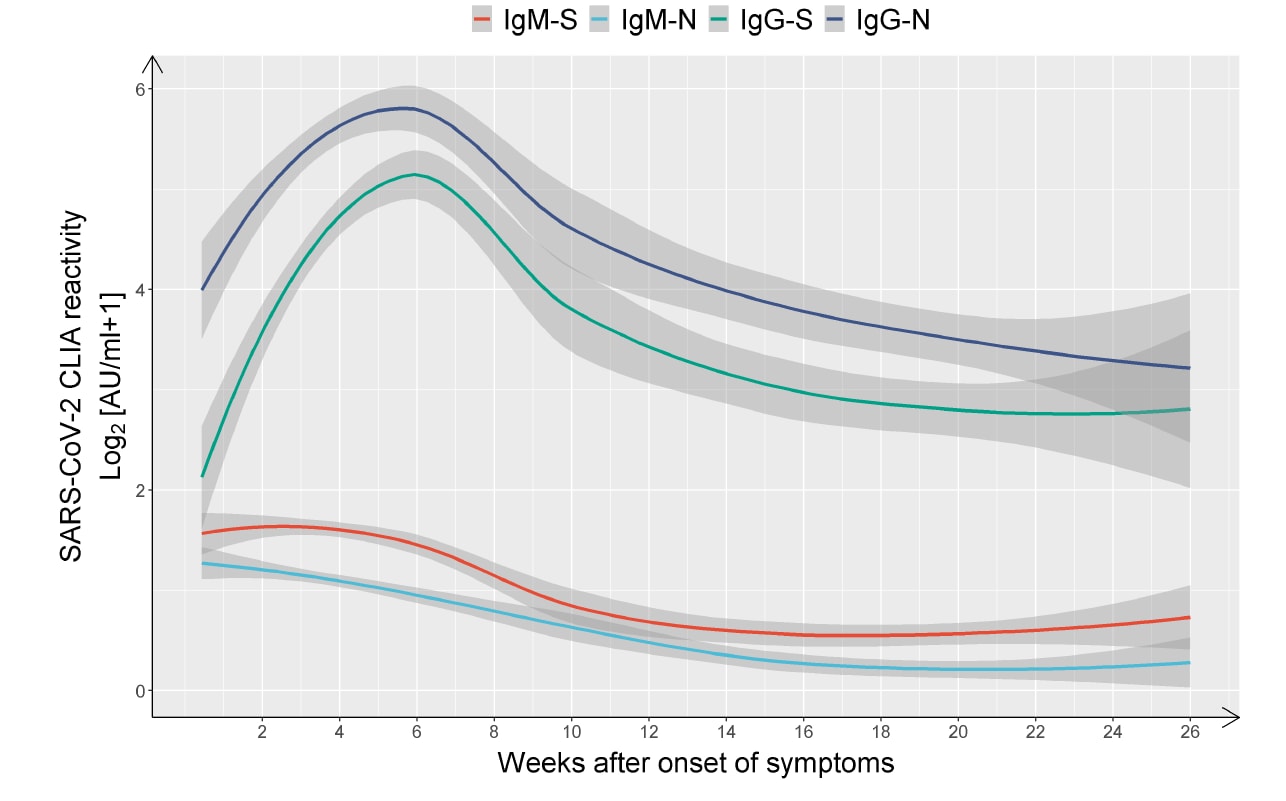
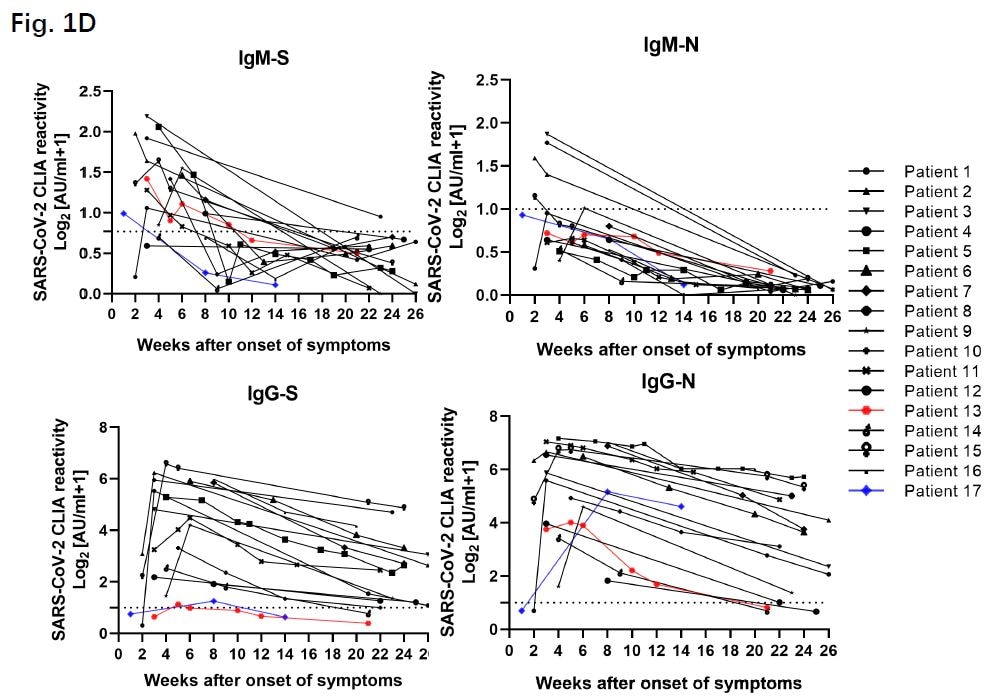
SARS-CoV-2 infection induces sustained humoral immune responses in convalescent patients following symptomatic COVID-19external icon. Wu et al. medRxiv (July 24, 2020).
Key findings:
- Symptomatic COVID-19 patients exhibit an early and rapid antibody response and maintain high levels of IgG that recognizes the spike protein (S) and the nucleocapsid protein (N) for at least 6 months after disease onset (Figure 1).
- 87% of patients had a detectable IgG-N at week 2 that remained elevated for the duration of the study.
- Among 17 patients with repetitive sampling, there was a rapid decline in IgM titers but a sustained IgG response (Figure 2).
- At 2-3 weeks after symptom onset, antibody levels were significantly higher among patients for whom SARS-CoV-2 RNA was no longer detected.
Methods: Serologic evaluation of 349 symptomatic patients for up to 26 weeks. Antibody titers of 17 patients with repetitive sampling were analyzed to confirm results of the larger study group. Antibody titers were compared by disease severity, gender, and age in a subset of patients (n = 209). Limitations: Smaller numbers for longitudinal data; limited to symptomatic patients.
Implications: Immunity among recovered symptomatic COVID-19 patients appears to be maintained for at least 6 months, contributed mostly by IgG S/N. Continued evaluation of the correlates of protection from SARS-CoV-2 infection over longer periods of time and among asymptomatic patients is needed.
Figure 1
Note: Adapted from Wu et al. Total cross-sectional data from 349 symptomatic patients with antibodies against the virus spike protein shown in red for IgM and green for IgG. Anti-nucleocapsid (N) antibodies are shown light blue for IgM, and dark blue for IgG. Log2 absorbance units per milliliter from a chemiluminescence assay (CLIA) are plotted on the Y-axis represent quantitative antibody levels. Licensed under CC-BY-NC-ND 4.0.
Figure 2
Note: Adapted from Wu et al. Sequential sampling and antibody (IgG and IGM) titers against the RBD of the spike protein (‘S’) and the nucleoprotein (‘N’) of SARS-CoV-2 from 17 symptomatic COVID-19 patients. Log2 absorbance units per milliliter from a chemiluminescence assay (CLIA) are plotted on the Y-axis represent quantitative antibody levels. The cut-off value for IgM and IgG detection is shown as a dotted line. Red and blue lines show two patients who had anti-N IgG responses though not anti-S. Licensed under CC-BY-NC-ND 4.0.
PEER-REVIEWED
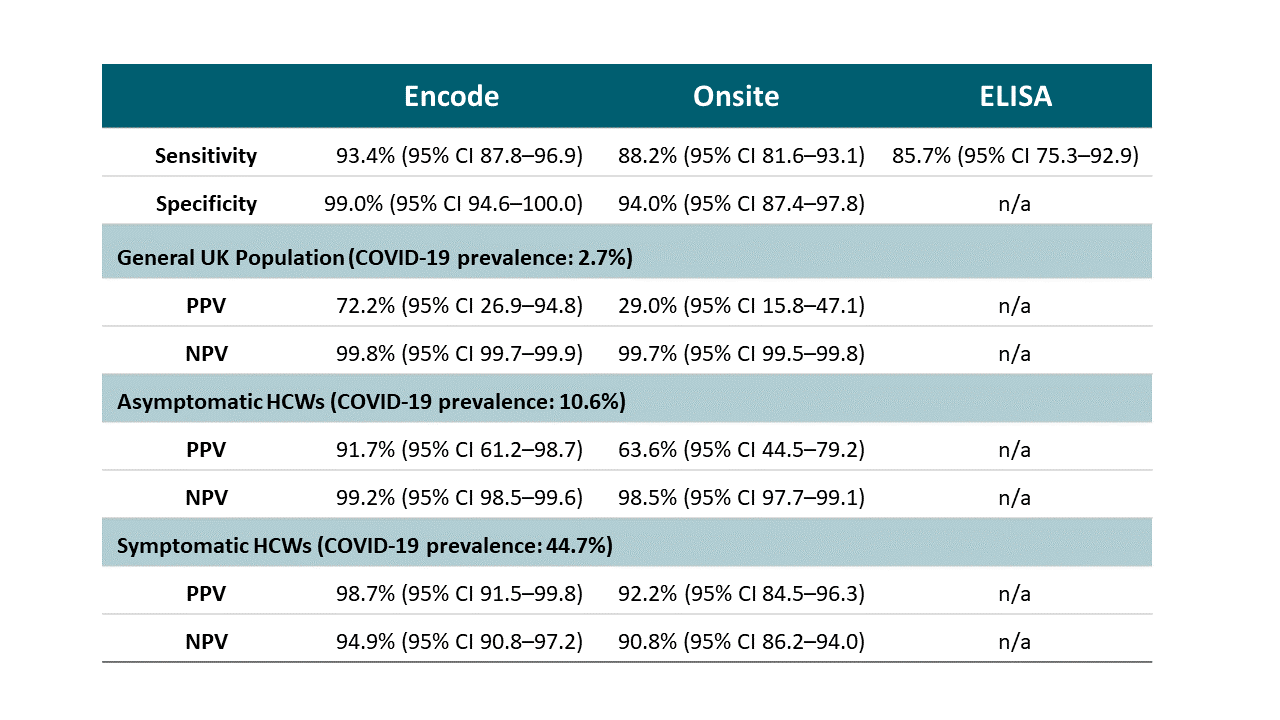
Point-of-care serological assays for delayed SARS-CoV-2 case identification among health-care workers in the UK: a prospective multicentre cohort studyexternal icon. Pallett et al. Lancet Respiratory Medicine (July 24, 2020; Correctionexternal icon on July 30, 2020).
Key findings:
- While one point-of-care (POC) serological assay displayed reasonable sensitivity (93.4%) and specificity (99.0%), the other POC assay had reduced sensitivity (88.2%) and specificity (94.0%) (Table).
- The laboratory serological assay also displayed reduced sensitivity (85.7%).
- The estimated seroprevalence of IgG for symptomatic healthcare workers (HCWs) was 44.7% (95% CI 42.0–47.4) and for asymptomatic HCWs was 10.6% (95% CI 7.6-13.6).
Methods: Two-part prospective study with a multicenter cohort of HCWs who delivered direct clinical care to SARS-CoV-2-infected patients and experienced mild-to-moderate COVID-19 symptoms with onset at least 14 days before serological testing, between April 8 and June 12, 2020. (1) Performance evaluation of 2 POC serological assays, Encode SARS-CoV-2 split IgM/IgG One Step Rapid Test Device (Zhuhai Encode Medical Engineering, Zhuhai, China) and Onsite CTK Biotech COVID-19 IgG/IgM Rapid Test (CTK Biotech, Poway, CA, USA), and one laboratory serological assay, EDI Novel Coronavirus COVID-19 IgG ELISA kit (Epitope Diagnostics, San Diego, CA, USA), compared with PCR for SARS-CoV-2 infection; RT-PCR testing was done where possible between days 1 to 7 from symptom onset. (2) Seroprevalence analysis using the POC serological assays among symptomatic (n = 1,299) and asymptomatic (n = 405) HCWs. Positive and negative predictive values based on test performance characteristics were calculated for use in symptomatic and asymptomatic HCWs and in the UK general population (based on a previously estimatedexternal icon 2.7% prevalence). Limitations: Only 2 point of care tests examined; laboratory immunoassay detected IgG to the nucleocapsid protein only and not to the spike protein or receptor binding domain; no information on targets used in POC assays.
Implications: POC tests to measure serologic response to SARS-CoV-2 infection could prove useful for serosurveillance, and performance of these tests needs to be properly assessed within intended populations. The low positive predictive values (<73%) of the POC tests examined here should be taken into account, and strategies to mitigate false positive results should be considered (see Peeling et al. The time to do serosurveys for COVID-19 is nowexternal icon. Lancet Respiratory Medicine).
Table:
Note: From Pallett et al. Performance of POC and laboratory serological assays.
PPV-positive predictive value; NPV-Negative predictive value; Encode- Encode SARS-CoV-2 split IgM/IgG One Step Rapid Test Device; Onsite- Onsite CTK Biotech COVID-19 split IgG/IgM Rapid Test; ELISA- EDI Novel Coronavirus COVID-19 IgG ELISA kit.
This article was published in Lancet Respiratory Medicine, Vol 8, Pallett et al., Point-of-care serological assays for delayed SARS-CoV-2 case identification among health-care workers in the UK: a prospective multicentre cohort study, Page 885-894, Copyright Elsevier 2020. This article is currently available at the Elsevier COVID-19 resource center: https://www.elsevier.com/connect/coronavirus-information-centerexternal icon.
- Liu et al. Size-dependent filtration efficiencies of face masks and respirators for removing SARS-CoV-2-laden aerosolspdf iconexternal icon. Infection Control and Hospital Epidemiology. Shows size-dependent filtration efficiencies for SARS-CoV-2-laden aerosols with different types of masks.
- Fennelly et al. Particle sizes of infectious aerosols: Implications for infection controlexternal icon. Lancet Respiratory Medicine. Reviews the literature on aerosols generated by persons with respiratory infections and calls for re-evaluating infection control guidelines to account for the predominance of small particles within infectious aerosols.
- Helleberg et al. Persistent COVID-19 in an immunocompromised patient temporarily responsive to two courses of remdesivir therapyexternal icon. Journal of Infectious Disease. Remdesivir could suppress but not eradicate SARS-CoV-2 infection in an immunocompromised patient with persistent COVID-19.
- Frazier et al. SARS-CoV-2 virus isolated from the mastoid and middle ear: Implications for COVID-19 precautions during ear surgeryexternal icon. JAMA Otolaryngology – Head & Neck Surgery. The presence of SARS-CoV-2 virus in the middle ear and mastoid prompts precautions during middle ear procedures.
- Kihira et al. Fatal cerebral infarct in a child with COVID-19external icon. Pediatric Radiology. Case study of a fatal infarction in a 5-year-old boy with COVID-19 presented to raise awareness that a post-infectious syndrome associated with SARS-CoV-2 can be fatal in children.
- Shook-Sa et al. Estimation without representation: Early SARS-CoV-2 seroprevalence studies and the path forwardexternal icon. Journal of Infectious Diseases. Discusses the pitfalls with using convenience samples and offers guidance for more representative and timely population estimates in seroprevalence studies.
- Smithgall et al. Laboratory testing of SARS CoV-2: A New York institutional experienceexternal icon. Advances in Molecular Pathology. Brief overview of the types of testing available for SARS-CoV-2 patient management and how testing has impacted the situation in New York City.
- Ozturker et al. Conjunctivitis as sole symptom of COVID-19: A case report and review of literatureexternal icon. European Journal of Ophthalmology. A case report of a health care worker diagnosed with COVID-19 who experienced conjunctivitis as only symptom of the disease.
- Qin et al. A post-COVID-19 effect: Increasing sport-related knee pain in adolescents after school reopeningexternal icon. SSRN. Increasing knee pain in adolescents after school reopening may be due to prolonged home stay resulting in physical decline and the rapidly increasing physical exercises after school reopening.
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.