COVID-19 Science Update released: July 10, 2020 Edition 29

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Here you can find all previous COVID-19 Science Updates.
Patient-level characteristics, such as obesity and diabetes, are associated with poorer outcomes among COVID-19 patients. However, the role of viral factors, including baseline viral load, on disease progression and outcome is not as well understood
PEER-REVIEWED
Impact of SARS-CoV-2 viral load on risk of intubation and mortality among hospitalized patients with coronavirus disease 2019. Magleby et al. Clinical Infectious Disease (June 30, 2020).
Key findings:
- Compared with having low viral load at admission, high viral load was independently associated with:
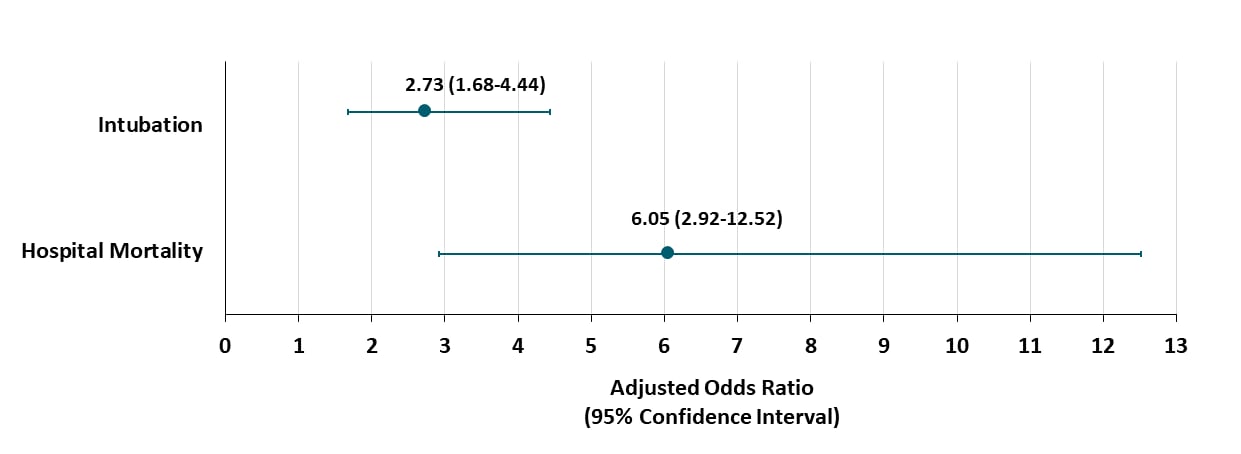
- In-hospital mortality (adjusted odds ratio [aOR] 6.05; 95% CI: 2.92-12.52).
- Intubation and mechanical ventilation (aOR 2.73; 95% CI: 1.68-4.44) (Figure).
Methods: Retrospective cohort study of 678 COVID-19 patients. SARS-CoV-2 viral load determined by Ct values from RT-PCR testing of NP swab at admission and categorized as high (Ct<25; n=220), medium (Ct 25-30; n=216), and low (Ct>30; n=242). Logistic regression adjusted for age, race, comorbidities, and clinical characteristics to examine the association of baseline viral load with in-hospital mortality and intubation. Limitations: Single NP swab at admission; retrospective data collection; only in-hospital mortality captured.
Figure:
Note: Adapted from Magleby et al. Association of SARS-CoV-2 baseline viral load (high; Ct<25 vs low; Ct>30) on hospital mortality or intubation. Multivariate analysis adjusted for age, race, comorbidities, and clinical characteristics.
PREPRINTS (NOT PEER-REVIEWED)
Association of SARS-CoV-2 genomic load with COVID-19 patient outcomes. Zacharioudakis et al. MedRxiv. (July 4, 2020). Publishedexternal icon in Annals of the American Thoracic Society (October 29, 2020).
Key findings:
- High SARS-CoV-2 viral load at hospital admission (compared with lower viral load) was associated with:
- Death, hospice care, mechanical ventilation or extracorporeal membrane oxygenation (aOR: 2.89; 95% CI: 1.7-14.5).
- Higher Charlson Comorbidity Index (predicts risk of death within 1 year of hospitalization for specific comorbidities), transplant recipient status, duration of illness (p<0.005) and higher pneumonia severity indices (p=0.03).
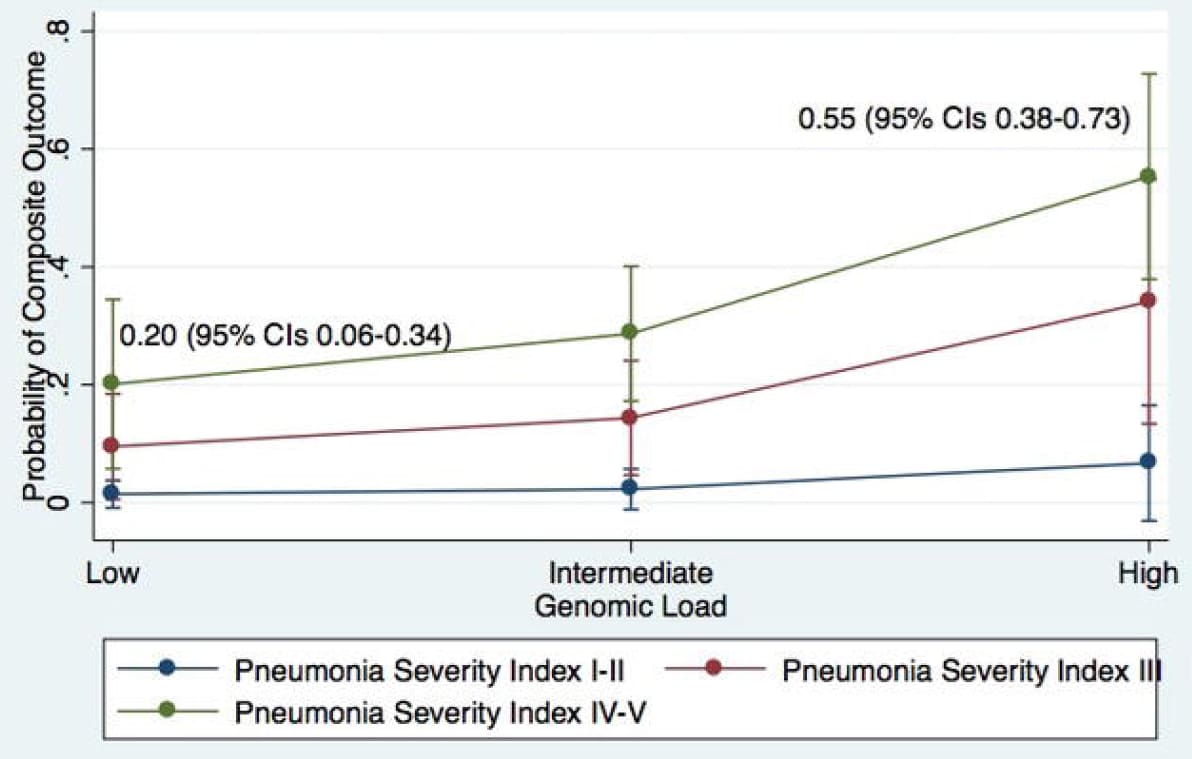
- Among patients with high pneumonia severity index, the robability of death, hospice care, mechanical ventilation or extracorporeal membrane oxygenation was higher for those with high viral loads compared with lower viral loads (Figure).
Methods: Cohort study of 316 hospitalized COVID-19 patients with clinical evidence of pneumonia. SARS-CoV-2 viral load measured at hospital admission by RT-PCR of NP swabs and defined as low (Ct ≥35), intermediate (Ct 25-35), and high (Ct≤ 25). Logistic regression adjusted for demographics, risk factors, comorbidities, and clinical characteristics was used to examine the association between baseline viral load and a composite outcome of death, hospice care, mechanical ventilation or extracorporeal membrane oxygenation. Limitations: Convenience sample; Ct values obtained at a single time point.
Figure:
Note: Adapted from Zacharioudakis et al. Estimated probability of death, hospital discharge to hospice, mechanical ventilation or extracorporeal membrane oxygenation by low, intermediate, and high viral load, stratified by pneumonia severity index.
Implications for 2 studies (Magleby et al & Zacharioudakis et al): SARS-CoV-2 viral load (reported as Ct values) may be a clinical marker to identify patients at high risk for adverse outcomes. Antiviral treatments that decrease viral load by inhibiting viral replication could improve outcomes.
Relying on the number of deaths officially attributed to COVID-19 might underestimate true mortality associated with COVID-19. Inadequate testing, deaths occurring outside of clinical care facilities, unrecognized non-respiratory manifestations of COVID-19 and deaths indirectly related to COVID-19 (e.g., inability to access care for other health concerns) can all lead to underestimates. Below, we summarize two reports that estimate excess deaths from all causes of mortality in the United States in early 2020, compared with expected deaths in that same time period.
PEER-REVIEWED
A. Estimation of excess deaths associated with the COVID-19 pandemic in the United States, March to May, 2020. Weinberger et al. JAMA Internal Medicine (July 1, 2020).
Key findings:
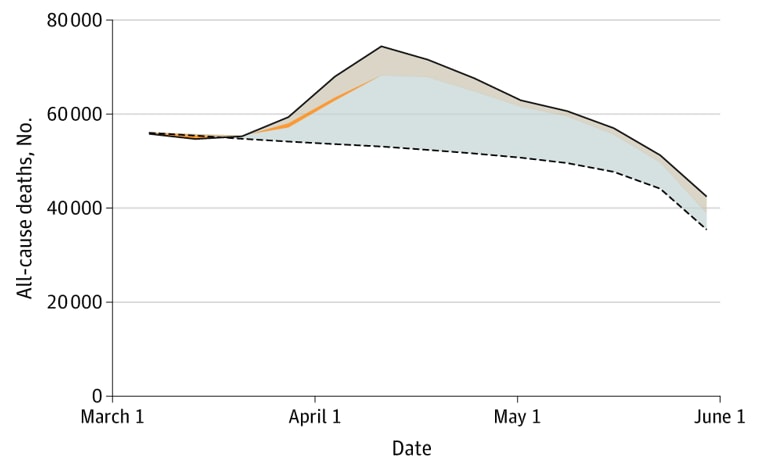
- An estimated 122,300 excess deaths occurred between March – May (Figure 1), with 95,235 (78%) attributed to COVID-19.
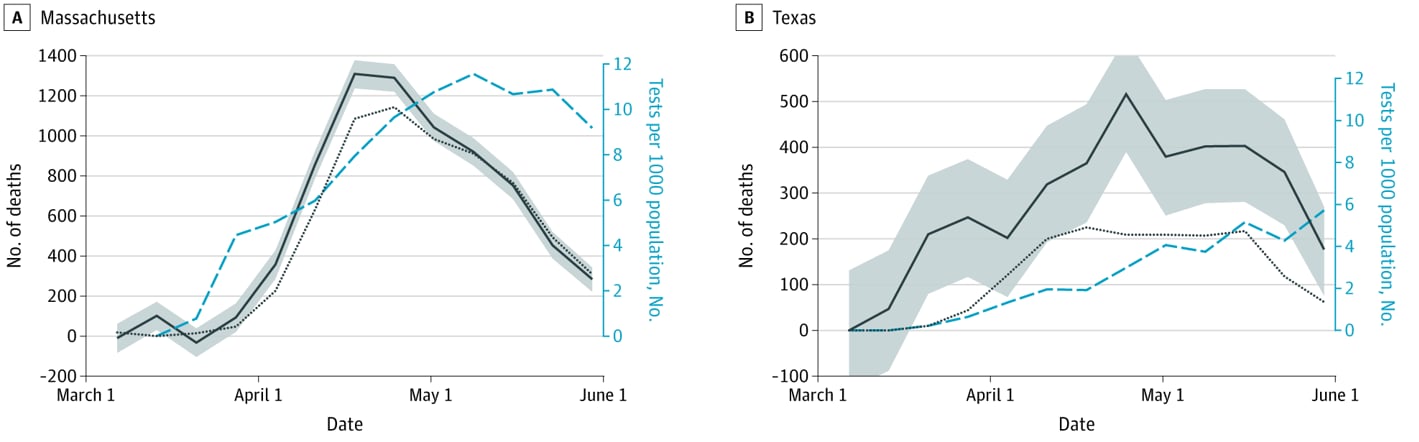
- The difference between estimated all-cause mortality and mortality attributed to COVID-19 varied by state and decreased as COVID-19 testing rates increased (Figure 2A and 2B).
Methods: Observational study comparing deaths recorded by the National Center for Health Statistics (NCHS) between March 1–May 30 2020 with expected deaths. Measurements included all deaths, deaths attributed to COVID-19, and deaths attributed to influenza and pneumonia. Expected deaths estimates were based on five years of previous NCHS data and adjusted for influenza, seasonality, year-to-year variation and reporting delays. Limitations: Missing data for Connecticut and North Carolina; finalized data not available at the time of analysis.
Figure 1
Note: Figure 1 from Weinberger et al. The solid line represents observed mortality, and the dashed line represents expected mortality based on estimates from NCHS data adjusted for reporting delays. The blue-gray area at the bottom represents mortality officially attributed to COVID-19, thin orange
area represents mortality attributed to influenza and pneumonia, and the beige area on top represents remaining excess mortality not attributed to COVID-19.
Figure 2
Note: Adapted from Weinberger et al. Trends in excess mortality in Massachusetts (A) and Texas (B) March─May, 2020. The solid black line represents observed mortality in each state; shaded areas are 95% prediction intervals. The dotted black line indicates mortality attributed to COVID-19 and the blue dashed line indicates the SARS-CoV-2 testing rate.
B. Excess deaths from COVID-19 and other causes, March-April, 2020. Woolf et al. JAMA (July 1, 2020).
Key findings:
- An estimated 87,001 excess deaths occurred March 1 ─ April 25, of which 56,246 (65%) were attributed to COVID-19.
- In states with highest COVID-19 morbidity, deaths from non-respiratory chronic conditions (e.g., diabetes) also increased, perhaps due to limited ability to seek care or non-respiratory manifestations of COVID-19.
Methods: Observational study comparing deaths recorded by NCHS between March 1 – April 25,2020 against expected deaths. Expected deaths were estimated based on NCHS data from 2013 – 2020 and adjusted for seasonality and year to year variation. Limitations: Missing data for Connecticut and North Carolina; finalized data not available at the time of analysis, no adjustment for reporting delays.
Implications for 2 studies (Weinberger et al. & Woolf et al.): Despite differences in time periods and analytic approaches, the two analyses indicate that mortality estimates based on official causes of death might underestimate true COVID-19-related mortality.
PEER-REVIEWED
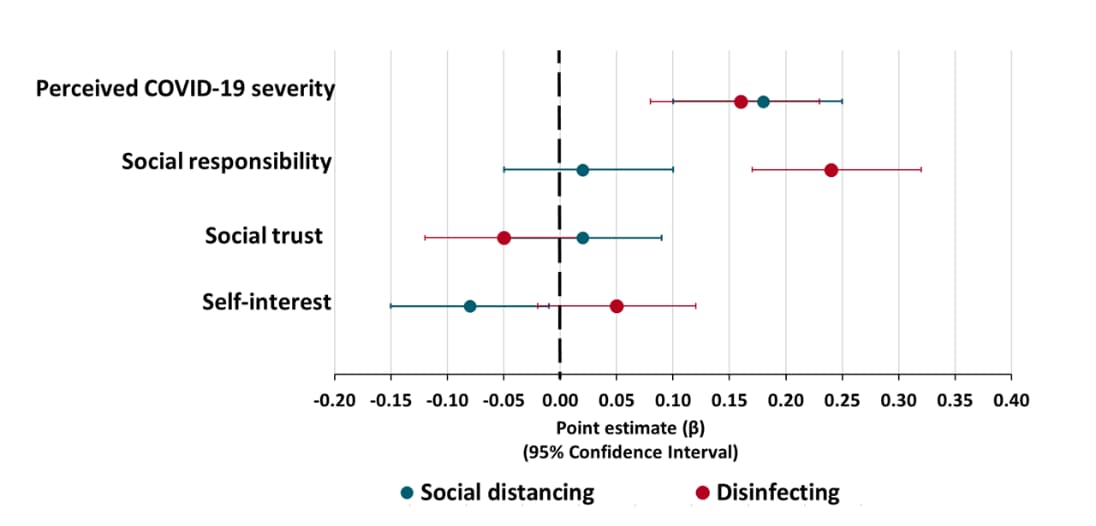
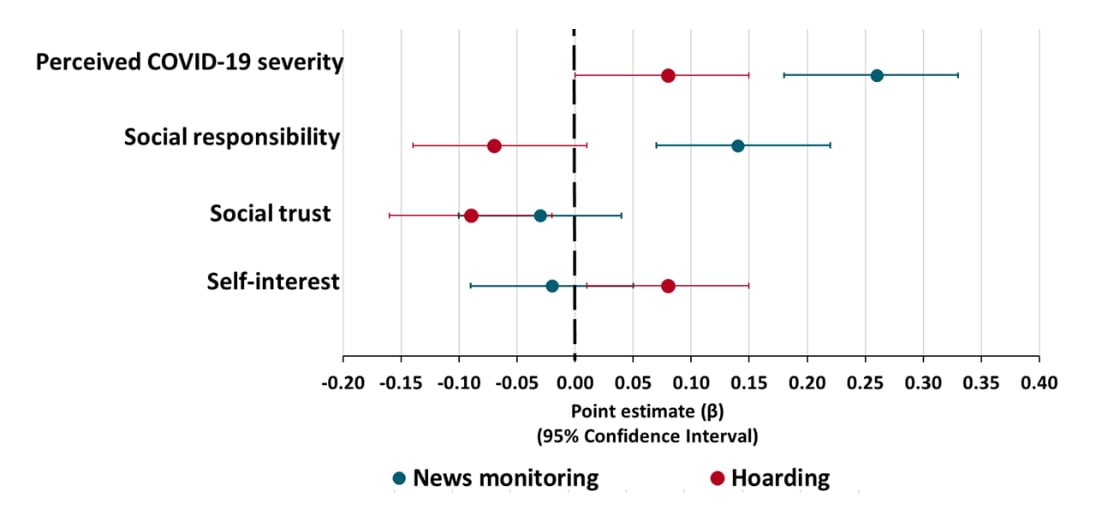
Attitudes and psychological factors associated with news monitoring, social distancing, disinfecting, and hoarding behaviors among US adolescents during the coronavirus disease 2019 pandemic. Oosterhoff et al. JAMA Pediatrics (June 29, 2020).
Key findings:
- Many adolescents reported monitoring COVID-19 news (89.4%) and disinfecting daily (87.8%); some reported hoarding (19.7%).
- Greater perceived severity of COVID-19 was associated with more social distancing, disinfecting, news monitoring, and hoarding (Figures 1 and 2).
- Greater social responsibility was associated with more disinfecting and news monitoring, but less hoarding (Figures 1 and 2).
- Greater self-interest was associated with less social distancing and more hoarding (Figures 1 and 2).
Methods: 770 adolescents (ages 13-18) completed an anonymous online survey from March 20-22, 2020 that assessed COVID-19–related experiences and attitudes, community attachments, and demographics. Multiple regression was used to analyze results. Limitations: Self-selected, cross-sectional, convenience sample (respondents were primarily white and female); timing of survey (future assessments may yield different results).
Implications: Messages emphasizing social responsibility and social trust may increase preventive health behavior in adolescents.
Figure 1
Note: Adapted from Oosterhoff et al. Point estimates with 95% confidence intervals of factors associated with adolescent social distancing (teal) and disinfecting (red) during the COVID-19 pandemic.
Figure 2
Note: Adapted from Oosterhoff et al. Point estimates with 95% confidence intervals of psychological factors associated with adolescent news monitoring (teal) and hoarding (red) during the COVID-19 pandemic.
PEER-REVIEWED
Novel use of home pulse oximetry monitoring in COVID-19 patients discharged from the emergency department identifies need for hospitalization. Shah et al. Academic Emergency Medicine (June 17, 2020).
Key findings:
- For patients discharged from the emergency department to home with a diagnosis of COVID and oxygen (O2) saturation >92%, developing a resting oxygen saturation <92% in COVID-19 was associated with:
- ARDS (RR: 8.2 [95% CI 1.7 – 38.7]).
- Septic shock (RR: 6.6 [95% CI 1.3 – 32.9]).
- 33% of non-hospitalized COVID-19 patients reported that home pulse oximetry reassured them and prevented a subsequent hospital visit.
Methods: Observational study of 77 RT-PCR SARS-CoV-2-positive patients with oxygen saturation >92% who were sent home with pulse oximetry. Univariate logistic regression was conducted to assess factors associated with a ARDS and septic shock. Limitations: No control group; small sample; single setting.
Implications: Home pulse oximetry monitoring in SARS CoV-2-infected patients may identify need for hospitalization and may prevent unnecessary emergency room visits.
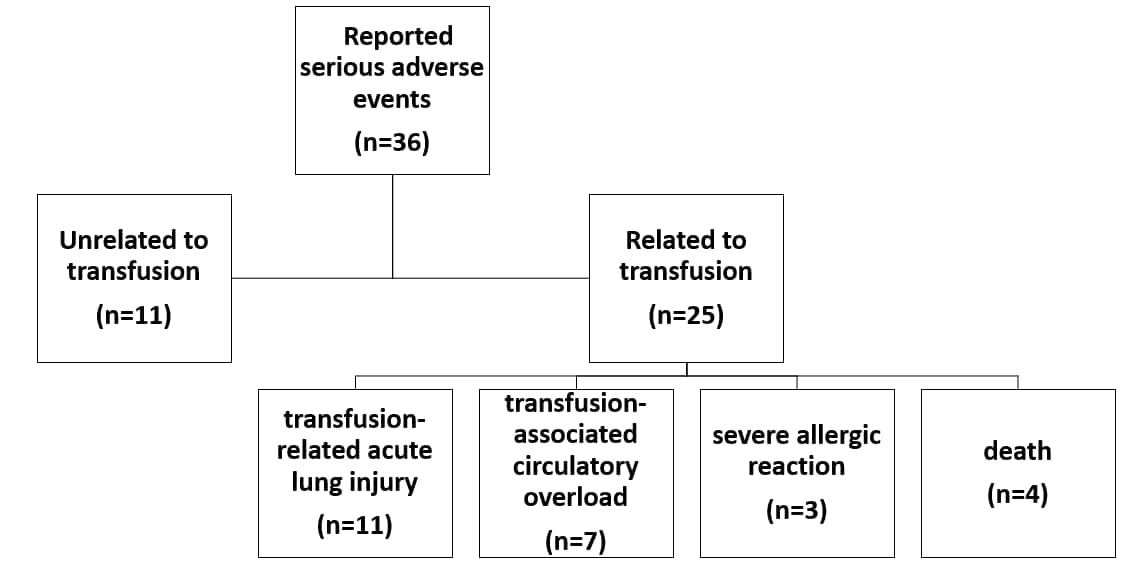
Early safety indicators of COVID-19 convalescent plasma in 5,000 patients. Joyner et al. The Journal of Clinical Investigation (June 11, 2020).
Key findings:
- Less than 1% of COVID-19 patients receiving convalescent plasma had a serious adverse event (SAE) in the first four hours after transfusion.
- Of 36 SAEs reported, 4 deaths and 21 non-death SAEs were related to the transfusion of COVID-19 convalescent plasma. (Figure).
Methods: Multicenter, open-label cohort of 5,000 hospitalized adults with COVID-19 with (n=4051) or at high risk of (n=949) severe or life-threatening disease. Patients received ABO-compatible COVID-19 convalescent plasma intravenously and were assessed for serious adverse events. Limitations: Broad inclusion criteria may result in heterogenous patient population; lack of detailed training on study protocol for study personnel and monitoring of sites owing to fast study start-up; varied research experience among participating sites.
Implications: Convalescent plasma for treatment in severely ill COVID-19 patients appears to be safe. Ongoing trials will be useful for understanding efficacy.
Figure:
Note: Adapted from Joyner et al. Serious adverse events among 5,000 hospitalized COVID-19 patients receiving intravenous ABO-compatible convalescent plasma. Four deaths judged as related (3 possibly, 1 probably). Relatedness determined by site investigators.
PEER-REVIEWED
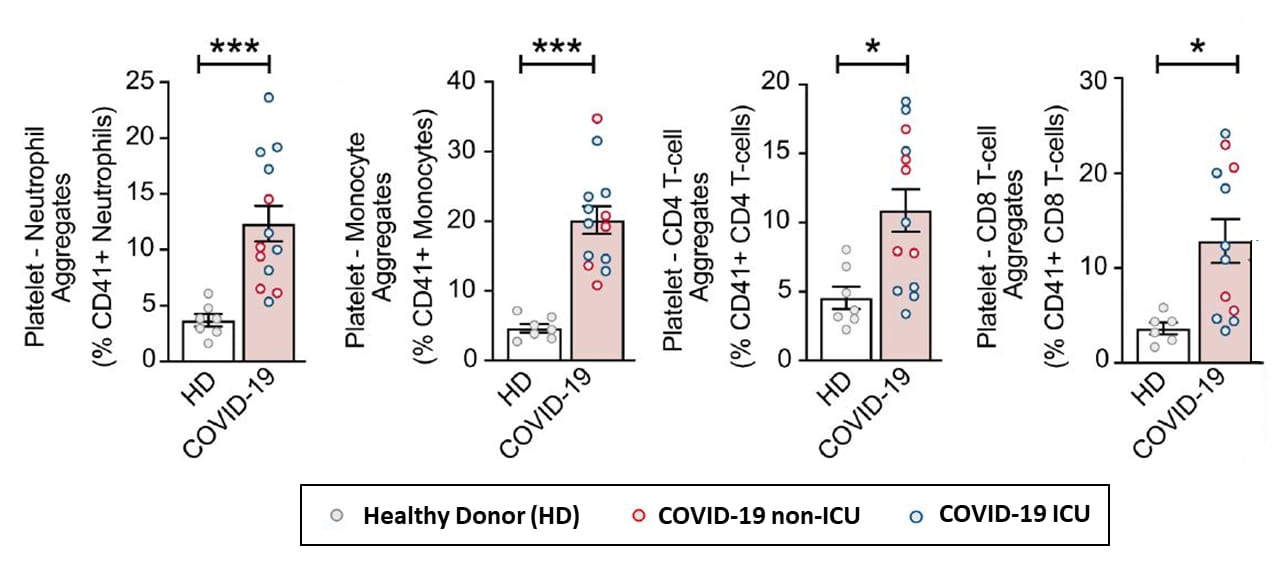
Platelet gene expression and function in COVID-19 patients. Manne et al. Blood (July 2, 2020)
Key findings:
- Platelets from COVID-19 patients display hyperreactivity.
- Platelet-neutrophil, platelet-monocyte, and platelet-T-cell aggregates (CD4+ and CD8+) were higher in COVID-19 patients compared with healthy donors (Figure 1).
- Thrombopoietin levels increased despite stable platelet counts.
- Hyperreactivity reduced when platelets were pre-treated with aspirin.
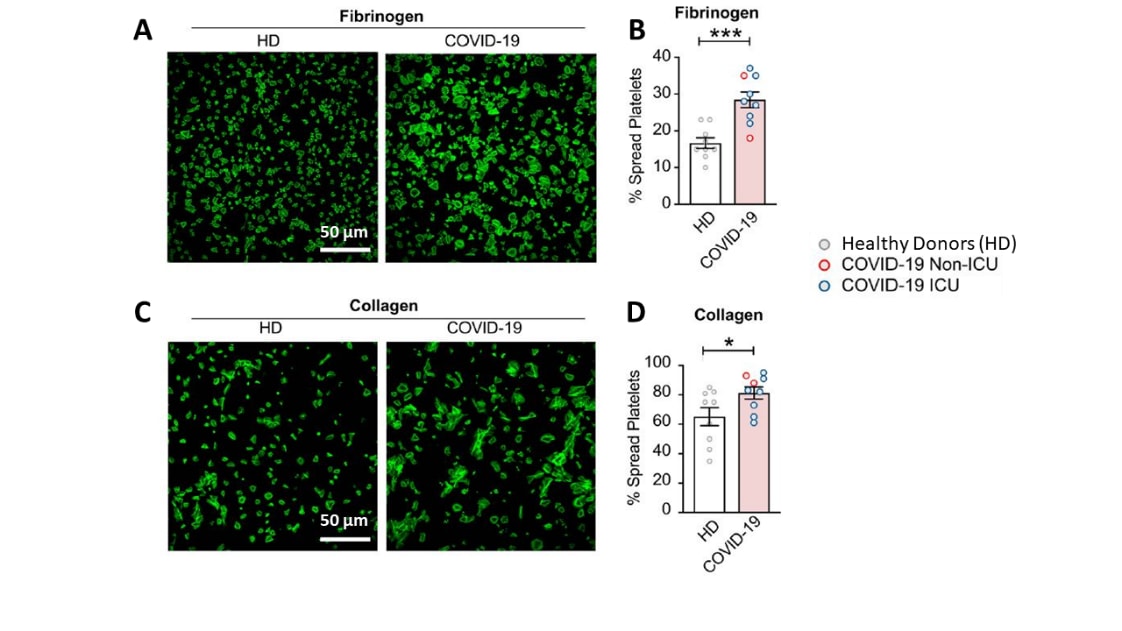
- Platelets also exhibited increased aggregation, adhesion, and spreading in COVID-19 patients compared with healthy donors (Figure 2).
Methods: Prospective clinical study comparing platelet responses in hospitalized COVID-19 patients (n=41), and age-, gender-, and race-matched healthy donors (n=41). Limitations: Small sample size; unable to examine clinical outcomes since no COVID-19 patients were diagnosed with thrombotic complications during study period.
Implications: SARS-CoV-2 infection increased platelet aggregation and hyperreactivity and this may contribute to COVID-19 disease outcomes including thrombotic events. Therapies modulating platelet activity may improve outcomes in COVID-19 patients.
Figure 1
Note: Adapted from Manne et al. Platelet-leukocyte aggregates in healthy donors (HD; white column bar) and COVID-19 patients (pink column bar). Among COVID-19 patients, blue dots indicate ICU patients while red dots indicate non-ICU patients *p<0.05, ***p<0.001.
Figure 2
Note: Adapted from Manne et al. Spread and adherence of platelets from healthy donors (HD) and COVID-19 patients. Platelets spread on fibrinogen and collagen for 30 mins (A and C) and spreading quantified (B and D). Among COVID-19 patients, blue dots indicate ICU patients while red dots indicate non-ICU patients *p<0.05, ***p<0.001
- VanderWeele, T. Challenges estimating total lives lost in COVID-19 decisions: Consideration of mortality related to unemployment, social isolation, and depression. JAMA. Discusses how estimates of excess deaths should consider mortality consequences of unemployment, isolation and depression.
- Malhame et al. The moral imperative to include pregnant women in clinical trials of interventions for COVID-19. Annals of Internal Medicine. Discusses why pregnant women must be included in COVID-19 clinical trials.
- Menza et al. Rapid uptake of home‑based HIV self‑testing during social distancing for SARS‑CoV2 Infection in Oregon. AIDS and Behavior. Pilot study of home HIV self-testing found high uptake and enthusiasm for home self-testing.
- Sinha et al. Is a “cytokine storm” relevant to COVID-19? JAMA Internal Medicine. Editorial provides a critical evaluation of the term cytokine storm, its relevance to COVID-19, and why the term may be misleading.
- Das Gupta et al. No more “social distancing” but practice physical separation. Canadian Journal of Public Health. Discussion of why the term social distancing is ambiguous and confusing for public health messaging.
- L’Huillier et al. Culture-competent SARS-CoV-2 in nasopharynx of symptomatic neonates, children, and adolescents. Emerging Infectious Diseases. Culture-competent virus isolated in vitro from 12 of 23 SARS-CoV-2–infected children, suggesting transmission from children is possible.
- Hall et al. Immune modulation in COVID-19: Strategic considerations for personalized therapeutic intervention. Clinical Infectious Diseases. Discussion of why a personalized, immunophenotype-driven approach to immunomodulation is important to explore.
- Merkler et al. Risk of ischemic stroke in patients with coronavirus disease 2019 (COVID-19) vs patients with influenza. JAMA Neurology. Cohort study found that rate of acute ischemic stroke among COVID-19 patients was higher than the rate among patients with influenza infection.
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.