COVID-19 Science Update released: April 9, 2021 Edition 84

The COVID-19 Science Update summarizes new and emerging scientific data for public health professionals to meet the challenges of this fast-moving pandemic. Weekly, staff from the CDC COVID-19 Response and the CDC Library systematically review literature in the WHO COVID-19 databaseexternal icon, and select publications and preprints for public health priority topics in the CDC Science Agenda for COVID-19 and CDC COVID-19 Response Health Equity Strategy.
Section headings in the COVID-19 Science Update align with the CDC Science Agenda for COVID-19.
Section headings in the COVID-19 Science Update have been changed to align
with the CDC Science Agenda for COVID-19.
Here you can find all previous COVID-19 Science Updates.
PEER-REVIEWED
Excess deaths from COVID-19 and other causes in the US, March 1, 2020, to January 2, 2021external icon. Woolf et al. JAMA (April 2, 2021).
Key findings:
- The US experienced 2,801,439 deaths, 22.9% more than expected (522,368 excess deaths).
- Deaths attributed to COVID-19 accounted for 72.4% of excess deaths in the US.
- Non-Hispanic Black persons comprised 16.9% of excess deaths but only 12.5% of the population.
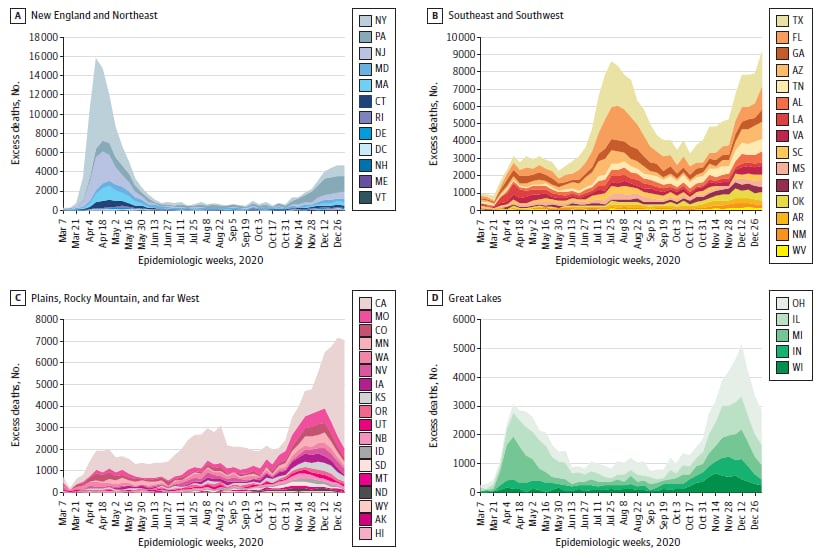
- Excess deaths surged in the Northeast and Great Lakes in the spring, followed by extended summer and early winter surges concentrated in Southern and Western states (Figure).
Methods: Modeled predicted expected deaths in 2020 based on US mortality data from 2014-2019 and compared with provisional, unweighted deaths reported from the District of Columbia and 49 states between March 1, 2020 and January 2, 2021. Limitations: Did not adjust for age changes in the population which could overestimate excess deaths; provisional data; inaccurate death certificates; model assumptions.
Implications: Earlier and more rigorous implementation of pandemic control measures might have prevented excess deaths in the US, particularly among minority populations. Excess deaths not attributed to COVID-19 could reflect mortality from undocumented COVID-19 infection or non-COVID-19 deaths due to secondary factors, such as disruptions in healthcare access caused by the pandemic.
Figure:
Note: Adapted from Woolf et al. Excess deaths by region (regions with similar patterns were combined), March 1, 2020 to January 2, 2021. Negative excess deaths were plotted as zero. Reproduced with permission from JAMA, 2021. Published online April 2, 2021. doi:10.1001/jama.2021.5199. Copyright© 2021 American Medical Association. All rights reserved.
COVID-19 case and mortality rates in the Federal Bureau of Prisonsexternal icon. Toblin et al. American Journal of Preventive Medicine (February 25, 2021).
Key findings:
- Bureau of Prison (BOP) incarcerated persons received proportionally more SARS-CoV-2 testing (50.3%) than the US population (32.5%), assuming one test per person.
- The crude case and mortality rates for incarcerated persons were 11,710.1 and 77.4 per 100,000 people, respectively.
- Compared with the U.S., the case ratio was 4.7, and standardized mortality ratio (SMR) was 2.6.
- The crude infection fatality rate (IFR) for both the BOP and US was 0.7%.
Methods: Data analysis from 146,036 incarcerated persons in the BOP inmate management system and its COVID-19 database, from February 29 to September 23, 2020. Compared BOP deaths with US COVID-19 deaths, and calculated age- and sex-adjusted SMR. Limitations: Pre-pandemic BOP population counts used for institution-specific testing rates; US testing rates and IFRs include children; SMR calculation did not control for underlying health conditions or racial/ethnic differences in incarcerated populations compared to the US population.
Implications: The high case rate in incarcerated persons relative to US adults might be attributed to close contact within congregate living conditions or higher testing rates. However, high rates of testing might have allowed BOP institutions to isolate cases and prevent further spread of COVID-19. Continued surveillance and adherence to mitigation strategies in correctional facilities could reduce the impact of COVID-19 in prison populations.
Factors associated with SARS-CoV-2 positivity in 20 homeless shelters in Toronto, Canada, from April to July 2020: a repeated cross-sectional studyexternal icon. Kiran et al. Canadian Medical Association Journal Open (March 30, 2021).
Key findings:
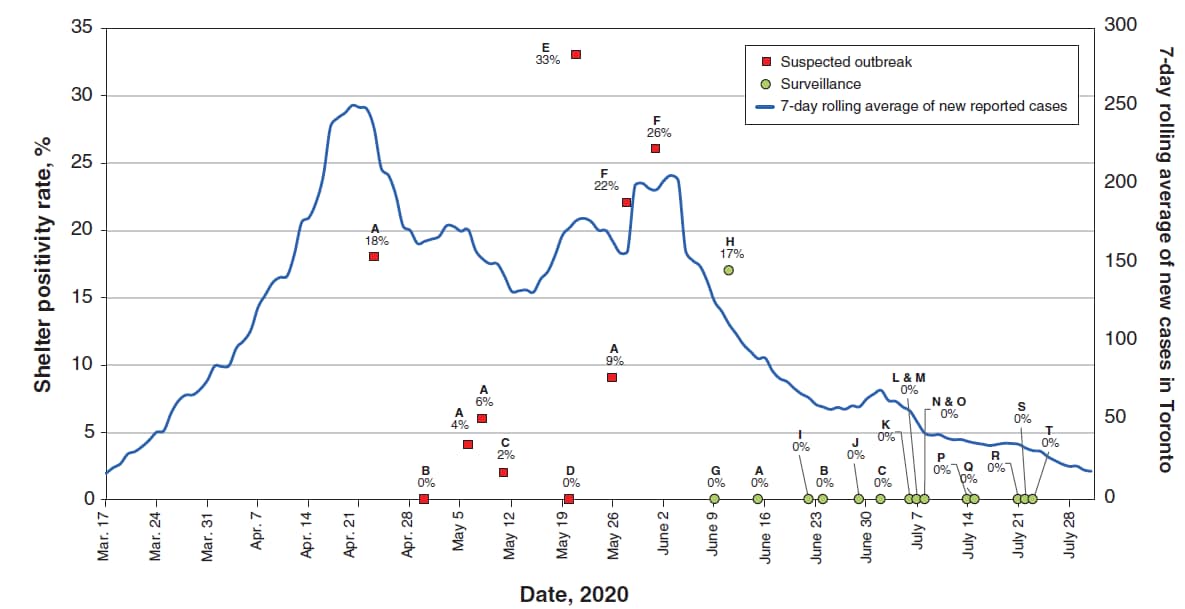
- Of 504 tests done because of a positive case at the same shelter, 69 (14%) were positive; of the 496 tests done for surveillance, 11 (2%) were positive.
- Among tests done for surveillance, only 1 of 17 shelters had any positive cases (Figure).
- No association was found between SARS-CoV-2 positivity and medical history or symptoms.
- Residents without a provincial health insurance card were more likely to test positive for COVID-19.
Methods: Cross-sectional retrospective chart audit of 872 residents for whom SARS-CoV-2 testing was done in 20 shelter locations in Toronto, Canada between April 1 and July 31, 2020. Compared the positivity rate for shelters where testing was conducted because of an outbreak (at least 1 known case) with those where testing was conducted for surveillance (no known cases). Limitations: Shelters selected for this study mostly served men; did not include homeless individuals who sleep outside or received testing at other facilities; potential selection bias.
Implications: When community SARS-CoV-2 case levels are elevated, routine testing in shelters might be warranted.
Figure:
Note: Adapted from Kiran et al. Shelter positivity rate for shelters where testing was conducted because of an outbreak and for shelters where testing was done for surveillance versus 7-day rolling average of new COVID-19 cases in Toronto. Letters refer to individual shelters. Licensed under CC-BY-NC-ND 4.0.
PEER-REVIEWED
Estimation of secondary household attack rates for emergent spike L452R SARS-CoV-2 variants detected by genomic surveillance at a community-based testing site in San Francisco.external icon Peng et al. Clinical Infectious Diseases (March 31, 2021).
Key findings:
- Prevalence of SARS-CoV-2 variants of concern B.1.427 and B.1.429—both carrying spike protein mutations S13I, W152C, and L452R—increased from 15.7% to 54.4% between November 2020 and January 2021.
- Adjusted relative risk (aRR) of infection after household exposure was marginally higher for B.1.429 (aRR = 1.36, 95% CI 1.01–1.83) compared to “non-West Coast variants” (strains other than B.1.427 and B.1.429).
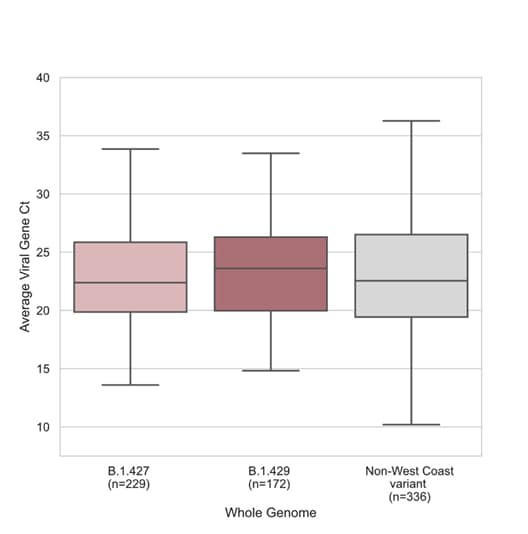
- Median SARS-CoV-2 RT-PCR cycle thresholds (Ct) were similar between persons infected with B.1.427, B.1.429 and non-West Coast variants (Figure).
Methods: SARS-CoV-2 genome sequences were recovered and analyzed from a community-based testing site and household contacts in San Francisco, CA. Used phylogenetic analysis to classify strains (n = 846) as circulating in the West Coast (B.1.427 and B.1.429) or other strains. Estimated household secondary attack rates and relative risks of infection (adjusted for age, ethnicity, household size and density). Limitations: Convenience sampling; prevalence of variants after January 2021 might be underestimated.
Implications: The prevalence of variants B.1.427 and B.1.429 increased sharply in a community-based sample of persons at risk for SARS-CoV-2 over a 3-month period. Maintaining prevention measures, like physical distancing and mask wearing, can reduce the elevated transmission risk of some emerging SARS-CoV-2 variants of concern. Genome sequencing can help detect and characterize these new strains and inform public action.
Figure:
Note: Adapted from Peng et al. SARS-CoV-2 RT-PCR Ct values for nasal swab samples from which whole high-quality genomes were recovered with B.1.427, B.1.429, and non-West Coast variants. Boxes and horizontal lines within them show interquartile ranges and medians. Licensed under CC-BY-NC-ND 4.0.
PREPRINTS (NOT PEER-REVIEWED)
Use of portable air cleaners to reduce aerosol transmission on a hospital COVID-19 wardexternal icon. Buising et al. medRxiv (March 31, 2021). Published in Infection, Disease & Healthexternal icon (November 2021).
Key findings:
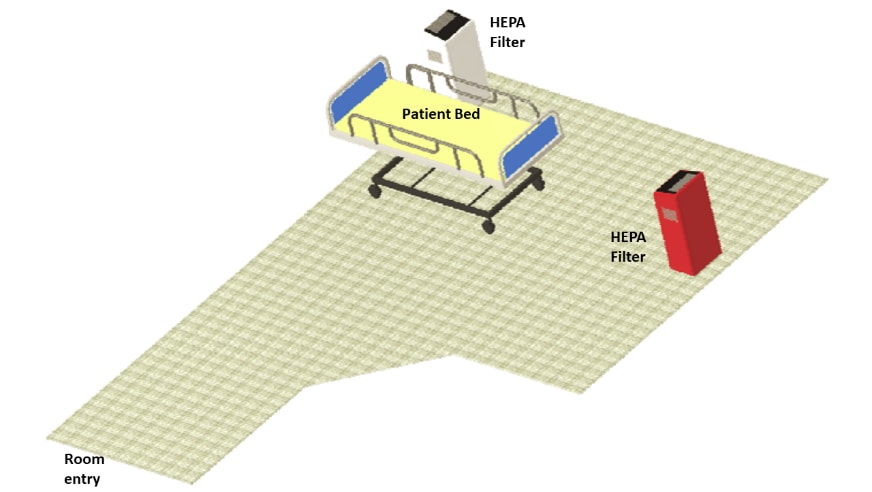
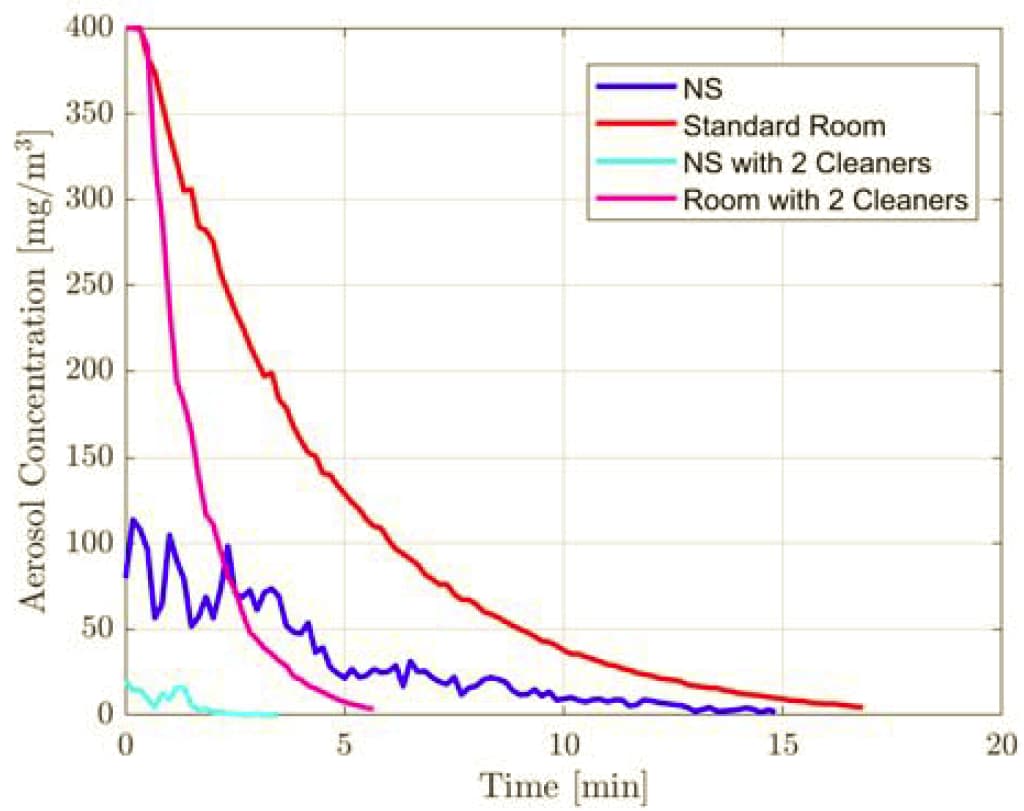
- Time to clear aerosols in a patient room was 5.5 minutes with mobile HEPA filters (Figure 1) compared to 16 minutes with existing ward HVAC system (Figure 2).
- Aerosols cleared in <3 minutes at the nurses’ station with mobile HEPA filters.
- There was little impact on aerosol clearance from opening the bathroom door in the patient room and keeping on the exhaust fan.
Methods: Observational experiment at the Royal Melbourne Hospital, Australia, from July to August 2020. Glycerine-based aerosol was used as a surrogate for respiratory aerosols in a single patient room and a nurses’ station. Time to clearance of 99% of aerosols was measured from inside the patient room and the nurses’ station with and without air cleaners (HEPA filters). Limitations: Study done in one hospital floor in one patient room; findings for SARS-CoV-2 might differ from those for glycerine-based aerosol.
Implications: Low-cost air cleaners in patient rooms and nurses’ stations may be a useful and cost-effective method in clinical spaces to reduce the risk of healthcare-acquired respiratory viruses that are transmitted via aerosols.
Figure 1:
Note: Adapted from Buising et al. Computer rendering of study set up for the single patient room with 2 HEPA filters. Licensed under CC-BY-NC-ND 4.0.
Figure 2:
Note: Adapted from Buising et al. Measured values for aerosol concentration over time from a patient room and a nurses’ station with usual heating ventilation and air conditioning, and from a patient room and a nurses’ station with 2 HEPA cleaners and closed door. Licensed under CC-BY-NC-ND 4.0.
PEER-REVIEWED
Currently authorized COVID-19 vaccines are not live vaccines and can be administered safely to people with immunosuppressive conditions or receiving immunosuppressive therapies. However, effectiveness of COVID-19 vaccines in this population is not known. Here we consider 2 studies evaluating immune responses to COVID-19 vaccines in immunocompromised individuals.
A. Immunogenicity of a single dose of SARS-CoV-2 messenger RNA vaccine in solid organ transplant recipients.external icon Boyarsky et al. JAMA (March 15, 2021).
Key findings:
- Only 17% (95% CI 14%-21%) of solid organ transplant recipients had antibody responses to one SARS-CoV-2 mRNA vaccine dose.
- Antibody responses were more likely after the mRNA-1273 (adjusted incidence rate ratio [aIRR] 2.15 [95% CI 1.29-3.57]) compared to the BNT162b2 vaccine.
- Compared to other patients, older patients (>60 years; aIRR 0.83 [95% CI 0.73-0.93]) and patients on anti–metabolite immunosuppression therapies (aIRR 0.22 [95% CI 0.15-0.34]) were less likely to develop responses.
Methods: Antispike protein antibody responses in 436 solid organ transplant recipients were assessed at a median of 20 days after the first dose of BNT162b2 or mRNA-1273. Limitations: Responses after the second vaccine dose and correlation of antibody responses to protection from SARS-CoV-2 infection are unknown.
B. Antibody response to a single dose of SARS-CoV-2 mRNA vaccine in patients with rheumatic and musculoskeletal diseases.external icon Boyarsky et al. Annals of the Rheumatic Diseases (March 23, 2021).
Key findings:
- Most patients (74% [95% CI 65%-81%]) with rheumatic and musculoskeletal (RMD) diseases had antibody responses to one SARS-CoV-2 mRNA vaccine dose.
- There were no reported differences by specific vaccine received.
- Patients receiving mycophenolate or rituximab were less likely to develop antibody responses compared to patients not receiving these therapies.
Methods: Antispike antibody responses in 126 RMD patients, most of whom were receiving therapies including non-biologic disease-modifying antirheumatic drugs (DMARDs, n = 23), biologic DMARDs (n = 17), corticosteroids (n = 4) or combinations (n = 45), were assessed at a median of 20 days after the first dose of either BNT162b2 or mRNA-1273. Limitations: Small sample; antibody responses assessed only after the first vaccine dose.
Implications for both studies (Boyarsky et al. A. and B.): Solid organ transplant recipients and patients receiving specific immunosuppressive therapies, including antimetabolites and rituximab, might have reduced antibody responses to one dose of SARS-CoV-2 mRNA vaccine compared to the general population. Further research is needed to determine whether people with immunosuppressive conditions or receiving specific immunotherapies would benefit from additional doses of COVID-19 vaccines or re-vaccination at a certain interval.
The effect of respiratory activity, non‐invasive respiratory support and facemasks on aerosol generation and its relevance to COVID‐19.external icon Wilson et al. Anaesthesia (March 30, 2021).
Key findings:
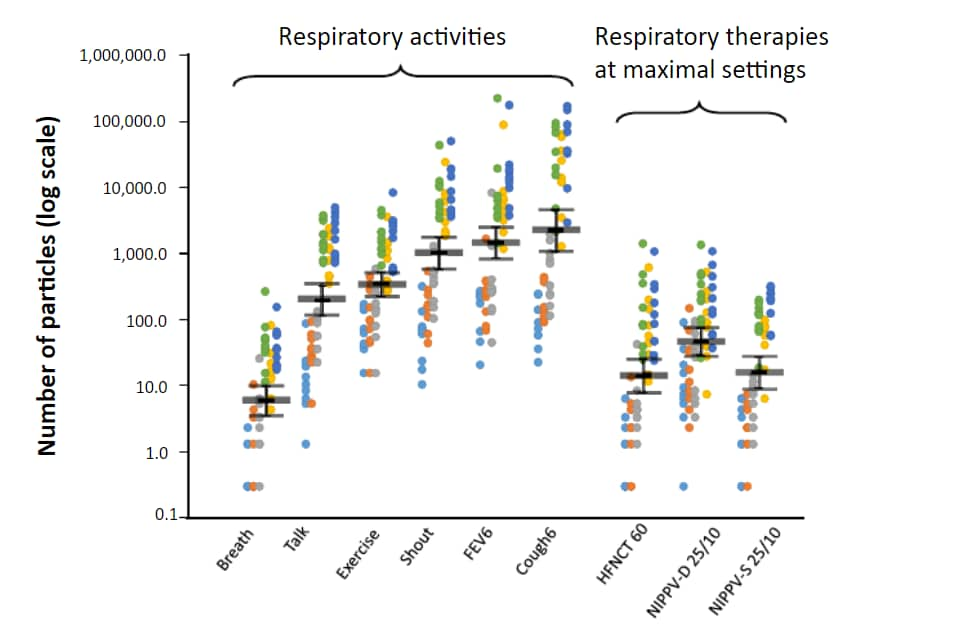
- Some common respiratory activities generated substantially more aerosols than non‐invasive respiratory therapies (Figure).
- Surgical face masks reduced aerosols during talking, shouting, forced expiratory volume (FEV) maneuvers and coughing.
Methods: Aerosols were measured in 10 healthy individuals during respiratory activities (quiet breathing; talking; exercise; shouting; FEV maneuvers; and coughing) with and without surgical face masks and three respiratory therapies (high‐flow nasal oxygen via cannula [HFNCT] and non‐invasive positive pressure ventilation via dual or single circuits [NIPPV-D, -S, respectively]). Limitations: Study was limited to a single experimental protocol and findings may not be generalizable.
Implications: This study highlights the importance of community masking to prevent transmission of SARS-CoV-2. Although aerosol exposure risk might be substantial when people have common physiologic responses to respiratory infection (e.g., coughing, labored breathing) or are engaged in everyday respiratory activities (e.g., shouting, exercising, or talking), use of a surgical face mask reduced aerosols.
Figure:
Note: Adapted from Wilson et al. Black bars show the mean and 95% CI of exhaled respiratory particles. Particle size ranges are: 10-25, 5-10, 3-5, 1-3, 0.7-1, and 0.5-0.7 µm. All respiratory therapies were conducted at the highest settings. FEV and cough were repeated six times. © 2021 Association of Anaesthetists. Used by permission of John Wiley & Sons.
Simulation-based estimation of SARS-CoV-2 infections associated with school closures and community-based nonpharmaceutical interventions in Ontario, Canada.external icon Naimark et al. JAMA Network Open (March 31, 2021).
Key findings:
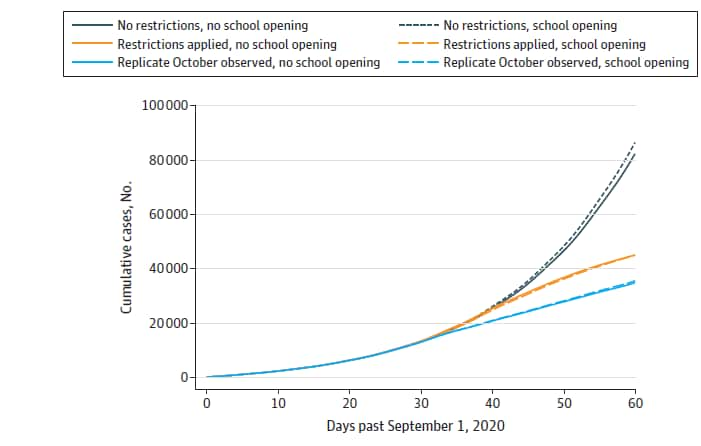
- Community-level nonpharmaceutical interventions (NPIs) focusing on reducing contacts and masking had a larger impact on incident and cumulative COVID-19 cases compared to closing schools and daycares (Figure).
- Simulations found >95% of new COVID-19 cases to be community acquired and <5% to be acquired within schools among students and teachers.
Methods: Agent-based model of two school/daycare reopening scenarios (do or do not reopen on 9/15/2020) with three community-level NPI scenarios (no additional NPI, NPI beginning in October 2020, and NPI limiting new infections to 0.8% per day-replicating October case data from Ontario), using a simulated population representative of Ontario. Limitations: Assumed schools have SARS-CoV-2 risk reduction measures in place; Ontario might not be representative of other areas.
Implications: Findings highlight the importance of prioritizing community-level NPI interventions over closing school and daycare to prevent SARS-CoV-2 transmission. Other recent studies (Bosslet et alexternal icon. and Bignami-van Assche et alexternal icon.) support implementing precautions, including physical distancing and face coverings, when opening schools.
Figure:
Note: Adapted from Naimark et al. Cumulative COVID-19 cases in Ontario from September 1, 2020-October 21, 2020 for 6 modeled scenarios: schools were open (dashed lines) or closed (solid lines) in the context of no community-wide restrictions (gathering restrictions, mask requirements, limiting workplace staff) or restrictions in hypothetical and previously reported scenarios. Licensed under CC BY.
Detection, Burden, and Impact
- Lindemer et al. Counties with lower insurance coverage are associated with both slower vaccine rollout and higher COVID-19 incidence across the United Statesexternal icon. medRxiv (Preprint, March 26, 2021). Published in Vaccines as Counties with Lower Insurance Coverage and Housing Problems Are Associated with Both Slower Vaccine Rollout and Higher COVID-19 Incidenceexternal icon (August 31, 2021). US counties with high levels of uninsured persons have significantly lower rates of COVID-19 vaccination, even after controlling for race and ethnicity, and had the highest COVID-19 incidence rate increases (comparing March 2021 to December 2020).
Transmission of SARS-CoV-2
- Paul et al. Pediatric household transmission of SARS-CoV-2 infectionexternal icon. medRxiv (Preprint, March 31, 2021). Published in JAMA Pediatrics as Association of Age and Pediatric Household Transmission of SARS-CoV-2 Infectionexternal icon (August 16, 2021). In 6,280 households in Ontario Canada, children aged 0-3 years or 4-8 years were more likely to transmit SARS-CoV-2 infection compared to children aged 14-17.
Natural History of SARS-CoV-2 Infection
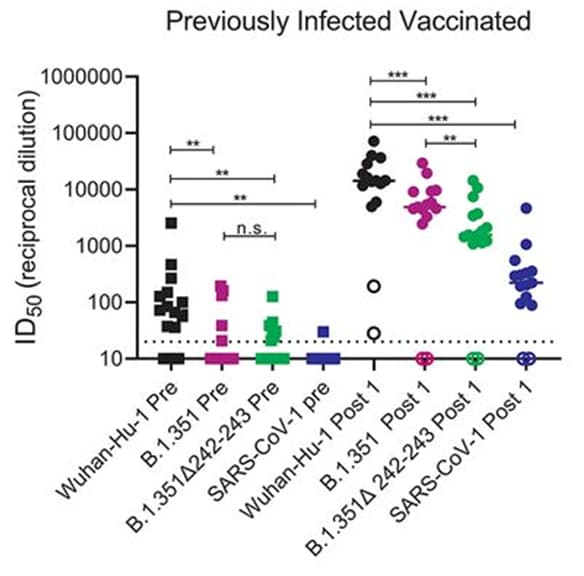
- Stamatatos et al. mRNA vaccination boosts cross-variant neutralizing antibodies elicited by SARS-CoV-2 infection.external icon Science (March 25, 2021). Sera from 15 previously infected, unvaccinated persons sporadically neutralized B.1.351, but 1 mRNA vaccine dose boosted cross-variant neutralizing titers against all B.1.351 variants by up to 1000-fold (Figure).
Figure:
Note: Adapted from Stamatatos et al. Serum dilution resulting in 50% neutralization (ID50) against WuHan-Hu-1, B.1.351, B.1.351Δ242-243, and SARS-CoV-1 pseudoviruses among recovered donors pre (squares) and post (circles) a single mRNA vaccine dose. Open circles show donors who were negative for anti-IgG RBD antibodies and RBD-specific IgG+ memory B cells prior to vaccination. Dashed line is lowest serum dilutions tested; ** p<0.01; *** p<0.001; n.s. not significant. Licensed under CC BY.
- Planas et al. Sensitivity of infectious SARS-CoV-2 B.1.1.7 and B.1.351 variants to neutralizing antibodiesexternal icon. Nature Medicine (March 26, 2021). Sensitivity of B.1.1.7 and B.1.351 variants were tested against sera from individuals infected with previously circulating strains or from those recently vaccinated. In both groups, neutralization was reduced against B.1.351 compared to B.1.1.7 and to D614G reference virus, indicating that B.1.351 might increase risk of infection even among immunized persons.
- Skidmore et al. Emergence of a SARS-CoV-2 E484K variant of interest in Arizonaexternal icon. medRxiv (Preprint, March 28, 2021). Variant B.1.243.1, which harbors the E484K mutation, has emerged in the United States and is becoming established in Arizona, New Mexico, and Texas.
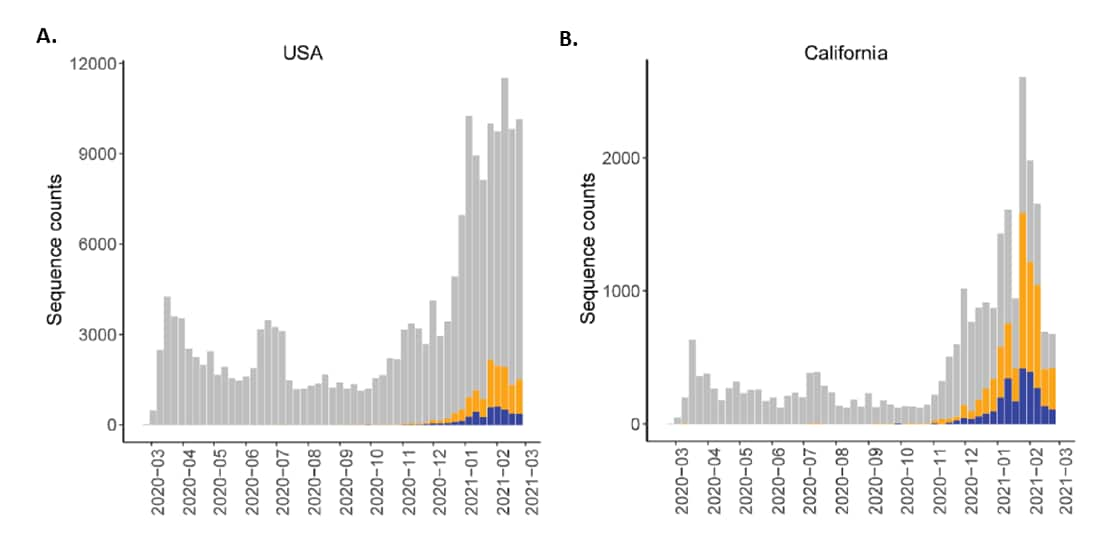
- McCallum et al. SARS-CoV-2 immune evasion by variant B.1.427/B.1.429external icon. bioRxiv (Preprint, April 1, 2021). Since December 2020, the number of B.1.427/B.1.429 genome sequences in the US and in California have increased rapidly, with a prevalence exceeding 50% in California in February 2021 (Figure). Neutralizing titers in plasma from vaccinated or convalescent individuals were reduced 3-6 fold against B.1.427/B.1.429 variants relative to wildtype pseudoviruses.
Figure:
Note: Adapted from MacCallum et al. Increasing prevalence of B.1.427/B.1.429 VOC lineages compared to overall sequence deposits in (A) the US, and in (B) California. Used by permission of authors.
Prevention, Mitigation, and Intervention Strategies
- Fox et al. The vaccine-elicited immunoglobulin profile in milk after COVID-19 mRNA-based vaccination is IgG-dominant and lacks secretory antibodies.external icon medRxiv (Preprint, March 26, 2021). Antibodies (Abs) elicited in breast milk >14 days post mRNA vaccination are IgG-dominant, with only 6 of 10 post-vaccine samples positive for IgA. In post-SARS-COV-2 infection milk antibody response, Spike-specific IgA is dominant and highly correlated with secretory Abs, which are resistant to enzymatic degradation in mucosae.
- Robbins et al. SARS-CoV-2 infection despite vaccination: an under-reported COVID-19 cohortexternal icon. Clinical Medicine Journal (March 2021). In this cross-sectional review of SARS-CoV-2 positive swabs from 174 non-ICU inpatients conducted in the UK in February 2021, 27 patients (mean age 82.3 years) had previously received a vaccine at a mean of 18.19 days before testing positive; 11/27 tested positive within 14 days of vaccination.
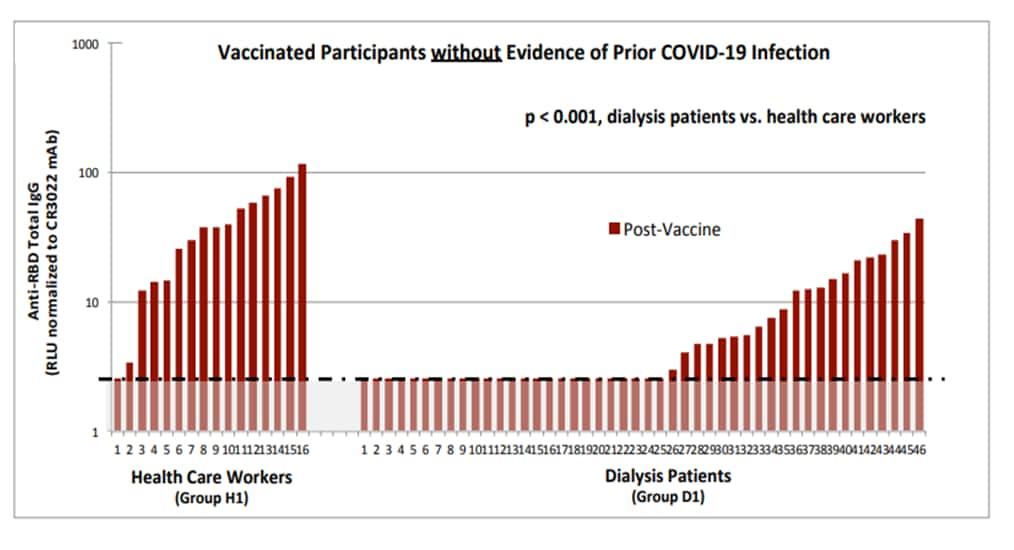
- Goupil et al. Short-term antibody response and tolerability of one dose of BNT162b2 vaccine in patients receiving hemodialysisexternal icon. medRxiv (Preprint, April 1, 2021). Published in Canadian Medical Association Journal as Short-term antibody response after 1 dose of BNT162b2 vaccine in patients receiving hemodialysisexternal icon (May 31, 2021). Hemodialysis patients tolerated the BNT162b2 vaccine well, but more frequently had a non-detectable antibody response (54%) compared to healthcare workers (6%) following vaccination (Figure).
Figure:
Note: Adapted from Goupil et al. IgG antibodies against the region binding domain (RBD) of the SARS-CoV-2 Spike protein 3 weeks after vaccination in health care workers (n = 16) and 4 weeks after vaccination in dialysis patients (n = 46). The dotted line represents the positive threshold for detection. Used by permission of authors.
Disclaimer: The purpose of the CDC COVID-19 Science Update is to share public health articles with public health agencies and departments for informational and educational purposes. Materials listed in this Science Update are selected to provide awareness of relevant public health literature. A material’s inclusion and the material itself provided here in full or in part, does not necessarily represent the views of the U.S. Department of Health and Human Services or the CDC, nor does it necessarily imply endorsement of methods or findings. While much of the COVID-19 literature is open access or otherwise freely available, it is the responsibility of the third-party user to determine whether any intellectual property rights govern the use of materials in this Science Update prior to use or distribution. Findings are based on research available at the time of this publication and may be subject to change.