2014-2015 Influenza Season Week 42 ending October 18, 2014
All data are preliminary and may change as more reports are received.
Synopsis:
During week 42 (October 12-18, 2014), influenza activity was low in the United States.
- Viral Surveillance: Of 8,412 specimens tested and reported by U.S. World Health Organization (WHO) and National Respiratory and Enteric Virus Surveillance System (NREVSS) collaborating laboratories during week 42, 403 (4.8%) were positive for influenza.
- Novel Influenza A Virus: One human infection with a novel influenza A virus was reported.
- Pneumonia and Influenza Mortality: The proportion of deaths attributed to pneumonia and influenza (P&I) was below the epidemic threshold.
- Influenza-associated Pediatric Deaths: Two influenza-associated pediatric deaths were reported, including one influenza-associated pediatric death that occurred during the 2013-2014 season.
- Outpatient Illness Surveillance: The proportion of outpatient visits for influenza-like illness (ILI) was 1.4%, which is below the national baseline of 2.0%. One region reported ILI above their region-specific baseline level. Puerto Rico experienced high ILI activity; Louisiana experienced low ILI activity; New York City and 49 states experienced minimal ILI activity and the District of Columbia had insufficient data.
- Geographic Spread of Influenza: The geographic spread of influenza in Guam was reported as widespread; Puerto Rico and five states reported local activity; the U.S. Virgin Islands and 36 states reported sporadic activity; and the District of Columbia and nine states reported no influenza activity.
| HHS Surveillance Regions* | Data cumulative since September 28, 2014 (week 40) | |||||||
|---|---|---|---|---|---|---|---|---|
| Out-patient ILI† | % positive for flu‡ | Number of jurisdictions reporting regional or widespread activity§ | 2009 H1N1 | A (H3) | A(Subtyping not performed) | B | Pediatric Deaths | |
| Nation | Normal | 3.9% | 1 of 54 | 7 | 267 | 405 | 392 | 1 |
| Region 1 | Normal | 1.6% | 0 of 6 | 0 | 8 | 8 | 4 | 0 |
| Region 2 | Normal | 1.1% | 0 of 4 | 1 | 17 | 17 | 11 | 0 |
| Region 3 | Normal | 1.7% | 0 of 6 | 0 | 16 | 16 | 69 | 0 |
| Region 4 | Normal | 8.0% | 0 of 8 | 0 | 56 | 290 | 246 | 1 |
| Region 5 | Normal | 2.1% | 0 of 6 | 3 | 28 | 15 | 12 | 0 |
| Region 6 | Normal | 4.3% | 0 of 5 | 2 | 32 | 26 | 69 | 0 |
| Region 7 | Normal | 1.9% | 0 of 4 | 0 | 12 | 15 | 9 | 0 |
| Region 8 | Normal | 1.2% | 0 of 6 | 0 | 15 | 5 | 6 | 0 |
| Region 9 | Normal | 3.2% | 1 of 5 | 1 | 27 | 27 | 19 | 0 |
| Region 10 | Normal | 5.5% | 0 of 4 | 0 | 56 | 4 | 7 | 0 |
*HHS regions (Region 1 CT, ME, MA, NH, RI, VT; Region 2: NJ, NY, Puerto Rico, US Virgin Islands; Region 3: DE, DC, MD, PA, VA, WV; Region 4: AL, FL, GA, KY, MS, NC, SC, TN; Region 5: IL, IN, MI, MN, OH, WI; Region 6: AR, LA, NM, OK, TX; Region 7: IA, KS, MO, NE; Region 8: CO, MT, ND, SD, UT, WY; Region 9: AZ, CA, Guam, HI, NV; and Region 10: AK, ID, OR, WA).
† Elevated means the % of visits for ILI is at or above the national or region-specific baseline
‡ National data are for current week; regional data are for the most recent three weeks
§ Includes all 50 states, the District of Columbia, Guam, Puerto Rico, and U.S. Virgin Islands
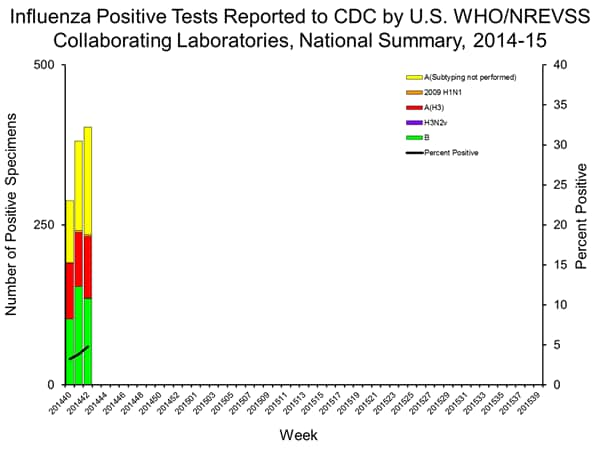
U.S. Virologic Surveillance:
WHO and NREVSS collaborating laboratories located in all 50 states, Puerto Rico, and the District of Columbia report to CDC the number of respiratory specimens tested for influenza and the number positive by influenza virus type and influenza A virus subtype. The results of tests performed during the current week are summarized in the table below. Region specific data are available at http://gis.cdc.gov/grasp/fluview/fluportaldashboard.html.
| Week 42 | |
|---|---|
| No. of specimens tested | 8,412 |
| No. of positive specimens (%) | 403 (4.8%) |
| Positive specimens by type/subtype | |
| Influenza A | 268 (66.5%) |
| 2009 H1N1 | 3 (1.1%) |
| H3 | 96 (35.8%) |
| Subytping not performed | 168 (62.7%) |
| Influenza B | 135 (33.5%) |
View National and Regional Level Graphs and Data | View Chart Data | View Full Screen | View PowerPoint Presentation
Novel Influenza A Viruses:
One human infection with a novel influenza A virus was reported by Wisconsin. The person was infected with an influenza A (H3N2) variant (H3N2v) virus, and has completely recovered from their illness. No ongoing human-to-human transmission has been identified and the case patient reported close contact with swine in the week prior to illness onset. This is the first H3N2v infection reported for the 2014-2015 influenza season, which began on September 28, 2014.
Early identification and investigation of human infections with novel influenza A viruses is critical in order to evaluate the extent of the outbreak and possible human-to-human transmission. Additional information on influenza in swine, variant influenza infection in humans, and strategies to interact safely with swine can be found at http://www.cdc.gov/flu/swineflu/h3n2v-case-count.htm.
Antigenic Characterization:
No antigenic characterization data is available for specimens collected after October 1, 2014.
Antiviral Resistance:
No antiviral resistance data is available for specimens collected after October 1, 2014.
The majority of currently circulating influenza viruses are susceptible to the neuraminidase inhibitor antiviral medications, oseltamivir and zanamivir; however, rare sporadic instances of oseltamivir-resistant 2009 H1N1 and A (H3N2) viruses have been detected worldwide. Antiviral treatment with oseltamivir or zanamivir is recommended as early as possible for patients with confirmed or suspected influenza who have severe, complicated, or progressive illness; who require hospitalization; or who are at high risk for serious influenza-related complications. Additional information on recommendations for treatment and chemoprophylaxis of influenza virus infection with antiviral agents is available at http://www.cdc.gov/flu/antivirals/index.htm.
Pneumonia and Influenza (P&I) Mortality Surveillance:
During week 42, 5.3% of all deaths reported through the 122 Cities Mortality Reporting System were due to P&I. This percentage was below the epidemic threshold of 6.0% for week 42.
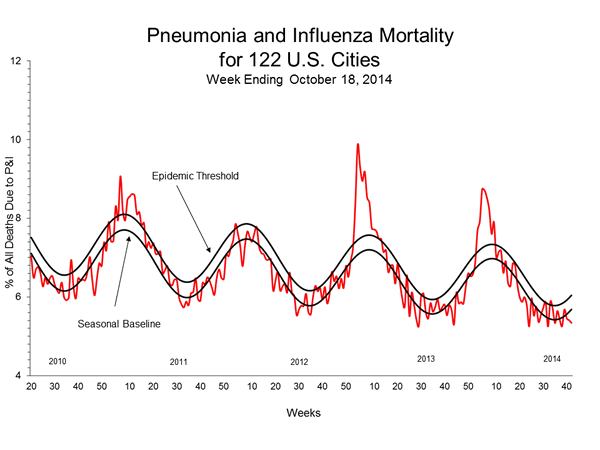
View Full Screen | View PowerPoint Presentation
For the 2014-2015 influenza season, CDC/Influenza Division and the National Center for Health Statistics (NCHS) are collaborating on a pilot project to use NCHS mortality surveillance data for the rapid assessment of pneumonia and influenza (P&I) mortality. To view the data, please click here (http://www.cdc.gov/flu/weekly/nchs.htm).
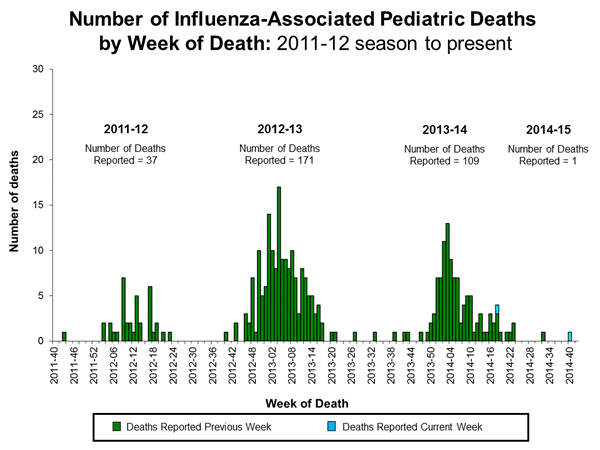
Influenza-Associated Pediatric Mortality:
Two influenza-associated pediatric deaths were reported to CDC during week 42. One death was associated with an influenza A (H3) virus and occurred during week 40 (week ending October 4, 2014). A total of one influenza-associated pediatric death has been reported during the 2014-2015 season.
One death was associated with an influenza B virus and occurred during the 2013-2014 season and brings the total number of reported pediatric deaths occurring during that season to 109.
Additional data can be found at: http://gis.cdc.gov/GRASP/Fluview/PedFluDeath.html.
Influenza-Associated Hospitalizations:
The Influenza Hospitalization Surveillance Network (FluSurv-NET) conducts all age population-based surveillance for laboratory-confirmed influenza-related hospitalizations in select counties in the Emerging Infections Program (EIP) states and Influenza Hospitalization Surveillance Project (IHSP) states. FluSurv-NET estimated hospitalization rates will be updated weekly starting later this season. Additional FluSurv-NET data can be found at: http://gis.cdc.gov/GRASP/Fluview/FluHospRates.html and http://gis.cdc.gov/grasp/fluview/FluHospChars.html.
Outpatient Illness Surveillance:
Nationwide during week 42, 1.4% of patient visits reported through the U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet) were due to influenza-like illness (ILI). This percentage is below the national baseline of 2.0%.
(ILI is defined as fever (temperature of 100°F [37.8°C] or greater) and cough and/or sore throat.)
Additional data are available at http://gis.cdc.gov/grasp/fluview/fluportaldashboard.html.
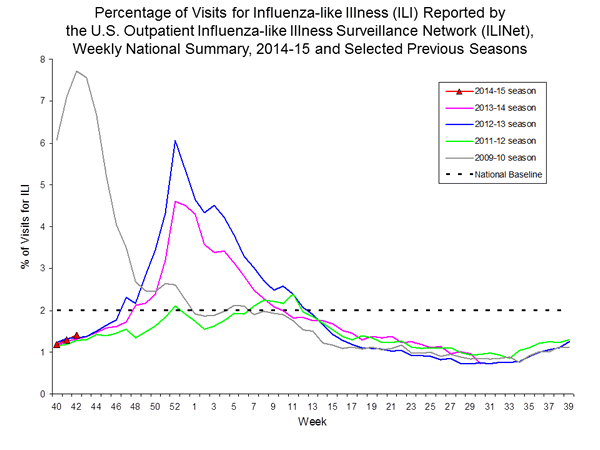
View National and Regional Level Graphs and Data | View Chart Data | View Full Screen | View PowerPoint Presentation
On a regional level, the percentage of outpatient visits for ILI ranged from 0.7% to 2.8% during week 42. One region reported ILI above their region-specific baseline level.
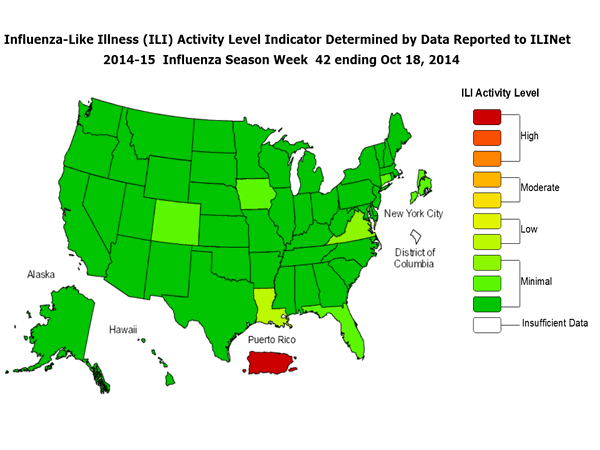
ILINet State Activity Indicator Map:
Data collected in ILINet are used to produce a measure of ILI activity* by state. Activity levels are based on the percent of outpatient visits in a state due to ILI and are compared to the average percent of ILI visits that occur during weeks with little or no influenza virus circulation. Activity levels range from minimal, which would correspond to ILI activity from outpatient clinics being below, or only slightly above, the average, to high, which would correspond to ILI activity from outpatient clinics being much higher than average.
During week 42, the following ILI activity levels were experienced:
- Puerto Rico experienced high ILI activity.
- Louisiana experience low ILI activity
- New York City and 49 states (Alabama, Alaska, Arizona, Arkansas, California, Colorado, Connecticut, Delaware, Florida, Georgia, Hawaii, Idaho, Illinois, Indiana, Iowa, Kansas, Kentucky, Maine, Maryland, Massachusetts, Michigan, Minnesota, Mississippi, Missouri, Montana, Nebraska, Nevada, New Hampshire, New Jersey, New Mexico, New York, North Carolina, North Dakota, Ohio, Oklahoma, Oregon, Pennsylvania, Rhode Island, South Carolina, South Dakota, Tennessee, Texas, Utah, Vermont, Virginia, Washington, West Virginia, Wisconsin, and Wyoming) experienced minimal ILI activity.
- Data were insufficient to calculate an ILI activity level from the District of Columbia.
*This map uses the proportion of outpatient visits to health care providers for influenza-like illness to measure the ILI activity level within a state. It does not, however, measure the extent of geographic spread of flu within a state. Therefore, outbreaks occurring in a single city could cause the state to display high activity levels.
Data collected in ILINet may disproportionally represent certain populations within a state, and therefore, may not accurately depict the full picture of influenza activity for the whole state.
Data displayed in this map are based on data collected in ILINet, whereas the State and Territorial flu activity map is based on reports from state and territorial epidemiologists. The data presented in this map is preliminary and may change as more data is received.
Differences in the data presented here by CDC and independently by some state health departments likely represent differing levels of data completeness with data presented by the state likely being the more complete.
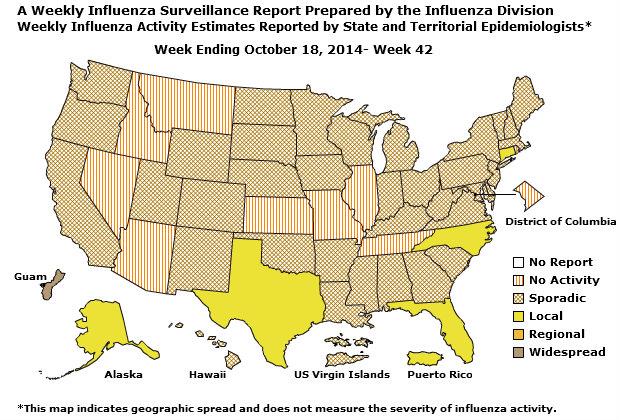
Geographic Spread of Influenza as Assessed by State and Territorial Epidemiologists
The influenza activity reported by state and territorial epidemiologists indicates geographic spread of influenza viruses, but does not measure the severity of influenza activity.
During week 42, the following influenza activity was reported:
- Widespread influenza activity was reported by Guam.
- Local influenza activity was reported by Puerto Rico and five states (Alaska, Connecticut, Florida, North Carolina, and Texas).
- Sporadic influenza activity was reported by the U.S. Virgin Islands and 36 states (Alabama, Arkansas, California, Colorado, Delaware, Georgia, Hawaii, Indiana, Iowa, Kentucky, Louisiana, Maine, Maryland, Massachusetts, Michigan, Minnesota, Mississippi, New Hampshire, Nebraska, New Jersey, New Mexico, New York, North Dakota, Ohio, Oklahoma, Oregon, Pennsylvania, South Carolina, South Dakota, Utah, Vermont, Virginia, Washington, West Virginia, Wisconsin, and Wyoming).
- No influenza activity was reported by the District of Columbia and nine states (Arizona, Idaho, Illinois, Kansas, Missouri, Montana, Nevada, Rhode Island, and Tennessee).