Collecting and Preparing Specimens for Anthrax Testing
Do not ship any specimens or samples without first obtaining authorization from both your state health department and CDC. Please contact CDC Emergency Operations Center at 1-770-488-7100 to coordinate anthrax testing and for further shipping instructions.
CDC accepts specimens from state public health laboratories and other federal agencies for analysis. Specimens from private healthcare providers and institutions must be submitted to the local state health department laboratory (state, county, city) for appropriate processing.
Specimen management activities at CDC include the receipt, categorization, and distribution of specimens from public health facilities to CDC laboratories for reference diagnostic testing and research studies. State-of-the-art technology is used to provide data and information summaries that are crucial in the assessment of public health trends and epidemic dynamics on a national scale.
How to collect specimens
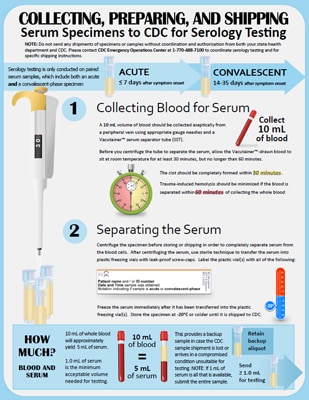
An acute serum sample for lethal factor (LF) toxin testing can be collected from 0 to 18 days after suspected exposure or the onset of symptoms. Earlier is preferred due to steady but gradual declines in toxin levels during antimicrobial therapy.
Serology testing will only be conducted in paired serum samples submitted at the same time, including an acute (≤7 days after symptom onset) and a convalescent-phase (14–35 days after symptom onset) specimen. It is recommended that the convalescent-phase specimen be taken 2 weeks after the acute specimen.
Collecting blood
- Collect 10 mL of blood aseptically from a peripheral vein using the appropriate gauge needles and a Vacutainer™ serum separator tube (SST); this will yield approximately 5 mL of serum.
- Before you centrifuge the tube to separate the serum, allow the Vacutainer™ drawn blood to sit at room temperature for at least 30 minutes, but no longer than 60 minutes.
- The clot should be completely formed within 30 minutes.
- Trauma-induced hemolysis should be minimized if the blood is separated within 60 minutes of collecting the whole blood specimens.
- For field collections where serum separation is not possible within 60 minutes of collection, store the specimen at 4°C using cold packs, and ship to a laboratory with centrifugation capabilities as soon as possible (ideally less than 24 hours).
Separating the serum
- Centrifuge the specimen before storing or shipping in order to completely separate serum from the blood cells.
- Centrifuge serum according to manufacturers’ instructions for the tube in use.
- After centrifuging the serum, use sterile technique to transfer the serum into a plastic freezing vial with a leak-proof screw-cap (e.g. 2 mL Sarstedt Screw Cap Micro Tube with an O-ring in the lid or equivalent).
- Only ship a portion of the 5mL serum specimen to CDC; retain one or more frozen aliquots as a “backup sample” in case the original shipment is lost or is unsuitable for testing upon arrival at CDC.
- 1 mL is the minimum acceptable volume needed by CDC. If 1 mL of serum is all that is available, please ensure that the entire sample is submitted.
- Label the plastic vial(s) with all of the following:
- Patient name and/or identification number
- Specimen collection date and time
- Whether the sample is an “acute” or a “convalescent” specimen
- Freeze the serum immediately after it has been transferred into the plastic freezing vial(s).
- Store the specimen at -20° C or colder until it is shipped to CDC.