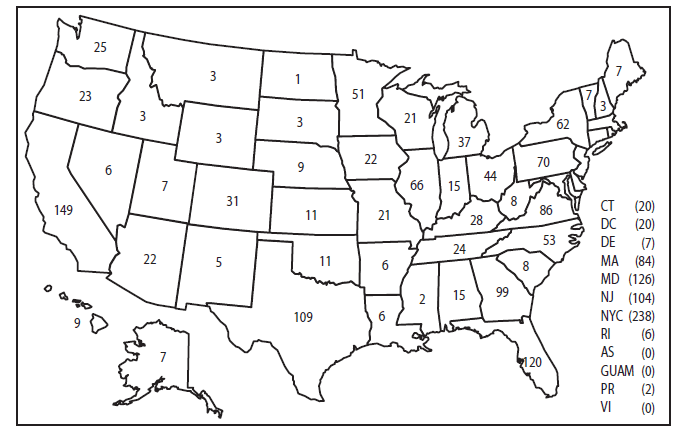
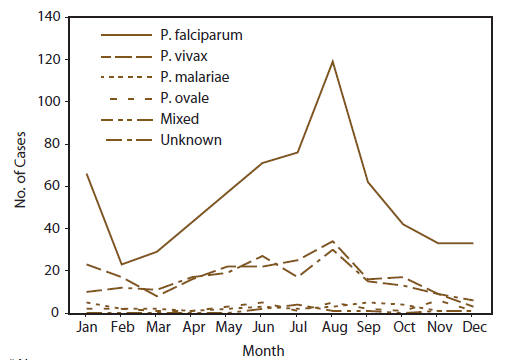
* The term United States includes all U.S. States and territories.
† Countries in west Africa include Benin, Burkina Faso, Cape Verde, Cote d'Ivoire, Gambia, Ghana, Guinea, Guinea-Bissau, Liberia, Mali, Mauritania, Niger, Nigeria, Senegal, Sierra Leone, and Togo.
§ Countries in south Asia include Afghanistan, Bhutan, India, Maldives, Nepal, Pakistan, and Sri Lanka (Ceylon).
 ShareCompartir
ShareCompartir