 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Meningococcal Disease -- New England, 1993-1998Neisseria meningitidis, a leading cause of bacterial meningitis and sepsis in children and young adults in the United States, causes both sporadic disease and outbreaks (1). Preventing and controlling meningococcal disease remains a public health challenge because of the multiple serogroups and the limitations of available vaccines (1,2). Vaccination with the polysaccharide meningococcal vaccine, which protects against serogroups A, C, Y, and W135 of N. meningitidis, is recommended by the Advisory Committee on Immunization Practices (ACIP) for controlling outbreaks but routine vaccination is not recommended for control of sporadic cases (1). During 1998, a cluster of meningococcal disease cases occurred in Rhode Island, and although the situation did not meet ACIP criteria for an outbreak (1), the Rhode Island Department of Health recommended vaccination of all residents aged 2-22 years. This action stimulated controversy in Rhode Island and the rest of New England (Connecticut, Maine, Massachusetts, New Hampshire, and Vermont) and prompted a review of the epidemiology of meningococcal disease in the region. This report describes meningococcal disease data reported to the region's state health departments during 1993-1998 and discusses the situation in Rhode Island. Surveillance. Connecticut and Massachusetts conducted prospective enhanced surveillance for meningococcal disease beginning in 1995 and 1996, respectively. In Rhode Island, additional case ascertainment was done in 1998 by reviewing hospital inpatient discharge data and hospital records for all confirmed and probable cases from 1992 through 1998. Surveillance in Maine, New Hampshire, and Vermont consisted of routine reporting for meningococcal disease. To calculate incidence, census data for 1996 were used. Case Definition and Detection Method. A confirmed case of meningococcal disease was defined as isolation of N. meningitidis from a normally sterile site (e.g., blood or cerebrospinal fluid [CSF]) from a person with clinically compatible illness. A probable case of meningococcal disease was defined as purpura fulminans or detection of meningococcal polysaccharide antigen in CSF in the absence of a diagnostic culture from a person with clinically compatible illness. Case Characteristics. During 1993-1998 in New England, 937 cases of meningococcal disease were reported. Of these, 899 (96%) met the definition for confirmed or probable meningococcal disease; 863 (96%) were confirmed by culture and 36 (4%) were probable cases. The proportion of confirmed cases varied by state from 100% (Vermont) to 84% (Rhode Island). Of the probable cases, 22 (61%) were reported as detection of meningococcal antigen in CSF, and 14 (39%) as purpura fulminans; 12 of 14 purpura fulminans cases were reported from Rhode Island. Of the 899 cases, 888 (99%) were considered primary (i.e., occurred in the absence of known close contact with another case-patient) (1). The median age of all case-patients was 17 years (range: 3 days-98 years); 455 (51%) were female, and 88 case-patients died (case fatality rate [CFR]=10%). The distributions of cases by age, sex, and serogroup were similar by state (Table 1). Rhode Island had a significantly higher CFR (21%) (p=0.001) than the other five states (Table 1). Ten (less than 1%) cases were associated with outbreaks; the remainder was classified as sporadic disease. Serogroups. Of the 758 (89%) cases with serogroup reported, 308 (41%) were serogroup C, 217 (29%) were serogroup Y, and 200 (26%) were serogroup B. Among case-patients with known serogroups, the proportion with serogroup Y meningococcal disease increased from 15% in 1993 to 43% in 1998 (pless than 0.005). Incidence. During 1993-1998, the average annual reported incidence of meningococcal disease was 1.1 cases per 100,000 population. Annual incidence increased significantly from 0.9 cases per 100,000 population in 1993 to 1.4 cases per 100,000 population in 1997 (chi square for linear trend, pless than 0.001) and declined from 1.4 to 0.9 cases per 100,000 population from 1997 to 1998 (pless than 0.001) (Figure 1). Excluding any state did not alter this trend. The lowest disease rate reported was 0.4 cases per 100,000 population (New Hampshire and Rhode Island in 1993) and the highest rate was 2.5 cases per 100,000 population (Rhode Island in 1997). Age groups with the highest incidence were children aged less than or equal to 2 years (6.4 cases per 100,000) and young adults aged 15-19 years (3.0 cases per 100,000). Reported by: RS Nelson, DVM, CA Morin, MPH, ML Cartter, MD, P Mshar, JL Hadler, MD, State Epidemiologist, Connecticut Dept of Public Health. G Beckett, MPH, K Gensheimer, MD, State Epidemiologist, Maine Dept of Human Services. J Isadore, MPH, P Kludt, MPH, BT Matyas, MD, A DeMaria, Jr, MD, State Epidemiologist, Massachusetts Dept of Public Health. M Johnson, J Greenblatt, MD, State Epidemiologist, New Hampshire Dept of Health and Human Services. L Mouradjian, P Nolan, MD, U Bandy, MD, State Epidemiologist, Rhode Island Dept of Health. JK Carney, MD, CJ Greene, MPH, PD Galbraith, DMD, State Epidemiologist, Vermont Dept of Health. Meningitis and Special Pathogens Br, Div of Bacterial and Mycotic Diseases, National Center for Infectious Diseases; Div of Applied Public Health Training, Epidemiology Program Office; and an EIS Officer, CDC. Editorial Note:Data in this report indicate that rates of meningococcal disease in New England increased during 1993-1997, then declined in 1998. The average annual rate in Rhode Island during this period was similar to rates in neighboring states. The rates also were similar to those reported in the United States during the same period (3,4). These changes in incidence probably represent natural fluctuations in disease incidence, changes in circulating strains of N. meningitidis, the population's susceptibility to disease, or some combination of these variables. Surveillance data indicated that the CFR among case-patients from Rhode Island were significantly higher than the CFR among case-patients from other states in the region. Twelve of 14 cases of purpura fulminans were reported from Rhode Island, and these case-patients had a higher CFR. However, when patients with purpura fulminans were eliminated from the analysis, the CFR in Rhode Island still remained elevated (20% versus 10%; pless than 0.003). Possible explanations for this difference in CFR include timing of antibiotic use and strain virulence. Some studies have reported that early antibiotic intervention is associated with reduced mortality (5); other studies have suggested that the finding may be attributable to confounding by variables such as host factors and severity of illness on presentation (6,7). In Rhode Island, case investigations have found that antibiotics were appropriately given, suggesting that other factors contributed to the high CFR. Between November 26, 1997, and February 23, 1998, Rhode Island reported nine confirmed cases (four serogroup C, three serogroup Y, and two serogroup B) and three probable cases of meningococcal disease, with three deaths. Although this cluster did not constitute an outbreak as defined by ACIP guidelines (1), a statewide vaccination program for residents aged 2-22 years was initiated. Approximately 60%-70% of the targeted population received the vaccine. The precedent of an earlier vaccination campaign in Woonsocket in 1996 and an increased reported incidence in disease and CFR generated public and medical concern and social and political pressure that influenced the decision to vaccinate (P.A. Nolan, MD, Rhode Island Department of Health, personal communication, 1998). Information on meningococcal disease in Rhode Island is available on the World-Wide Web at http://www.health.state.ri.us/meningoc.htm.* Although some cases may be prevented by this approach, its overall impact may be limited for several reasons: it will not protect children aged less than 2 years, in whom rates of disease are highest; it does not protect against serogroup B disease, which accounts for 26% of disease in the region; and, because the vaccine does not affect carriage, it will not affect disease among the 30%-40% of the target population who chose not to be vaccinated. Monitoring of disease in Rhode Island over the next few years will allow further evaluation of this strategy. During 1993-1998, less than 1% of cases in New England were classified as outbreak associated. Most cases of meningococcal disease were sporadic and therefore not preventable with strategies that target outbreaks. For efficacious protection of meningococcal disease in infants and young children, conjugate serogroup A, C, Y, and W135 meningococcal vaccines have been developed through methods similar to those used for Haemophilus influenzae type b conjugate vaccines (8,9). These vaccines will be used routinely in the United Kingdom within the next year (10) and should be available in the United States within 2-4 years. Until they become available, strategies to control meningococcal disease should continue to focus on antimicrobial chemoprophylaxis of close contacts and use of meningococcal polysaccharide vaccines as recommended by ACIP (1). References
* References to sites of nonfederal organizations on the World-Wide Web are provided as a service to MMWR readers and do not constitute or imply endorsement of these organizations or their programs by CDC or the U.S. Department of Health and Human Services. CDC is not responsible for the content of pages found at these sites. Table 1 Note: To print large tables and graphs users may have to change their printer settings to landscape and use a small font size.
TABLE 1. Cases of meningococcal disease, by number of cases, demographic
characteristics of patients, number and percentage of deaths, and serogroup -- New
England, 1993-1998
===============================================================================================================
Serogroup*
-------------------------------------------
Median Male Died B C Y
age ------------ ------------ ------------- ------------ ------------
State (yrs) No. (%) No. (%) No. (%) No. (%) No. (%)
-------------------------------------------------------------------------------------------------------------
Connecticut (n=231) 18.0 111 (48%) 19 ( 8%) 37 (18%) 81 (39%) 76 (37%)
Maine (n=80) 13.0 39 (49%) 6 ( 8%) 20 (30%) 32 (49%) 9 (13%)
Massachusetts (n=394) 17.0 199 (51%) 35 ( 9%) 97 (29%) 132 (40%) 94 (28%)
New Hampshire (n=81) 18.0 41 (51%) 9 (11%) 18 (28%) 29 (45%) 16 (25%)
Rhode Island (n=77) 15.0 36 (47%) 16 (21%) 17 (30%) 25 (45%) 13 (23%)
Vermont (n=36) 17.0 17 (47%) 3 ( 8%) 11 (32%) 9 (27%) 9 (27%)
New England (n=899) 17.0 417 (49%) 88 (10%) 200 (26%) 308 (41%) 217 (29%)
-------------------------------------------------------------------------------------------------------------
* Culture-confirmed cases. The proportion with other serogroups are included in the denomi-
nator.
===============================================================================================================
Return to top. Figure 1 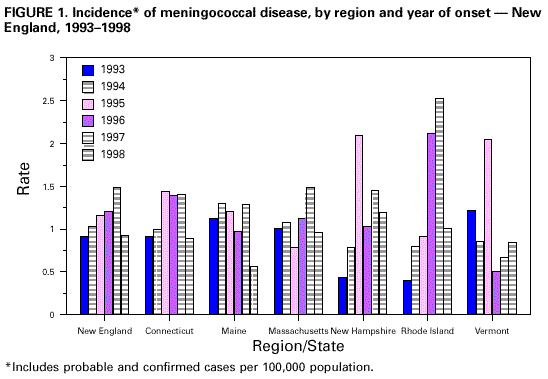 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 7/29/99 |
|||||||||
This page last reviewed 5/2/01
|