 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Update: Influenza Activity - United States and Worldwide, 1998-99 Season, and Composition of the 1999-2000 Influenza VaccineIn collaboration with the World Health Organization (WHO), the WHO international network of collaborating laboratories, and state and local health departments, CDC conducts surveillance to monitor influenza activity and to detect antigenic changes in the circulating strains of influenza viruses. This report summarizes surveillance for influenza in the United States and worldwide during the 1998-99 influenza season and describes the composition of the 1999-2000 influenza vaccine. United States Influenza activity began to increase in mid-January 1999 and peaked during the weeks ending February 6 through February 27. The predominant virus was influenza A(H3N2), although influenza type B viruses also circulated widely and were reported in all nine influenza surveillance regions. Influenza A(H1N1) viruses were sporadically isolated during the season in six of nine regions. During the weeks ending February 6 through February 27, 1999, greater than 40 state and territorial epidemiologists reported widespread or regional influenza activity*, with widespread activity first reported from a state during the week ending January 16 and reported last during the week ending April 10. Beginning the week ending January 23, the proportion of patient visits to U.S. influenza sentinel physicians attributed to influenza-like illness (ILI) increased above baseline levels (0-3%) to 4% and remained elevated for 7 consecutive weeks. The proportion of visits for ILI was at baseline levels in all surveillance regions by the week ending March 20. From October 4, 1998, through May 1, 1999, WHO and National Respiratory and Enteric Virus Surveillance System collaborating laboratories in the United States tested 86,826 specimens for respiratory viruses; 12,993 (15%) were positive for influenza. Of these, 10,041 (77%) were influenza type A, and 2952 (23%) were influenza type B. Of the 2501 subtyped influenza A viruses, 2481 (99%) were type A(H3N2), and 20 (1%) were type A(H1N1) ( Figure 1 ). Beginning the week ending January 30, the proportion of deaths attributed to pneumonia and influenza (P&I) reported by 122 U.S. cities exceeded the epidemic threshold** for 12 consecutive weeks. During the week ending March 13, the proportion of deaths attributed to P&I peaked at 8.8% ( Figure 2 ). Of the 327 U.S. A(H3N2) isolates collected from October 4 through May 1 and antigenically characterized at CDC, 295 (90%) were similar to the 1998-99 A(H3N2) vaccine strain, A/Sydney/5/97, and 32 (10%) had antigenically drifted from A/Sydney/5/97 based on hemagglutination inhibition testing. Six U.S. influenza A(H1N1) isolates were characterized as A/Bayern/7/95-like viruses, antigenically distinct from A/ Beijing/262/95, the 1998-99 A(H1N1) vaccine strain; however, the 1998-99 A(H1N1) vaccine strain produced high titers of antibodies that cross-react with A/Bayern/7/95. All 180 antigenically characterized B isolates were similar to the recommended type B vaccine strain, B/Beijing/184/93. Worldwide During October 1998-April 1999, influenza A(H3N2) viruses predominated in Austria, Bulgaria, Canada, China, Croatia, the Czech Republic, Denmark, Finland, France, Germany, Hong Kong, Iran, Israel, Japan, Korea, Latvia, Norway, Portugal, Romania, Russia, Slovakia, Sweden, Ukraine, and United Kingdom. Influenza A(H3N2) isolates also were reported from Algeria, Argentina, Australia, Belarus, Brazil, Ecuador, Egypt, French Guiana, Greece, Guam, Hungary, India, Italy, Malaysia, Martinique, the Netherlands, Nepal, Peru, the Philippines, Poland, Saudi Arabia, Senegal, Singapore, South Africa, Spain, Switzerland, Taiwan, and Thailand. Influenza A(H1N1) viruses were isolated from sporadic cases in China, Croatia, France, Hong Kong, Japan, Korea, Martinique, the Philippines, Portugal, Russia, Slovakia, South Africa, Spain, and Thailand. During outbreaks in Peru in February and March, influenza A(H1N1) was the most frequently isolated influenza virus type/ subtype. Other countries reporting influenza type A viruses include Belgium, Iceland, Lithuania, and Yugoslavia. Influenza B isolates predominated in Belarus, Hungary, Poland, Spain, and Taiwan. The number of influenza type B isolates was approximately equal to the number of influenza type A isolates in Belgium, Italy, the Netherlands, and Switzerland. Outbreaks associated with influenza type B viruses were reported in Brazil, French Guiana, and Japan. Influenza B viruses also were reported in Australia, Austria, Bulgaria, Canada, Chile, China, Croatia, the Czech Republic, Denmark, Finland, France, Germany, Greece, Guam, Hong Kong, Iceland, Israel, Latvia, Lithuania, Martinique, Nepal, Norway, Portugal, Romania, Russia, Saudi Arabia, Singapore, Slovakia, South Africa, Sweden, Thailand, United Kingdom, and Yugoslavia. In April 1999, the first two cases of human influenza A(H9N2) illness were identified among children hospitalized in March in Hong Kong, Special Administrative Region, People's Republic of China. Case-patients were girls, aged 1 and 4 years; both recovered from their illnesses. Investigations are under way in Hong Kong to determine the potential impact of this new subtype in humans. Surveillance in Hong Kong has been maintained at enhanced levels since human influenza A(H5N1) infections were identified in 1997. An additional five suspected human cases of H9N2 illness from Guangdong Province, China, were reported in March 1999 (1). No human cases of influenza A(H5N1) illness have been identified since December 1997. Composition of the 1999-2000 Vaccine The Food and Drug Administration's Vaccines and Related Biologic Products Advisory Committee (VRBPAC) recommended that the 1999-2000 trivalent vaccine for the United States contain A/Sydney/5/97-like(H3N2), A/Beijing/262/95-like(H1N1), and B/Beijing/184/93-like viruses. This recommendation was based on antigenic analyses of recently isolated influenza viruses, epidemiologic data, and postvaccination serologic studies in humans. Most influenza A(H3N2) isolates were A/Sydney/5/97-like viruses. A small percentage were distinguishable antigenically by hemagglutination-inhibition testing. However, these viruses were heterogeneous, and antigenic and genetic analysis did not reveal the emergence of a representative variant. Therefore, A/Sydney/5/97 will be retained as the influenza A(H3N2) 1999-2000 vaccine component. A/Beijing/262/95-like (H1N1) viruses were identified in Asia and South America and A/Bayern/7/95-like (H1N1) viruses were identified in Europe and the United States during the preceding year. Persons who were vaccinated in an experimental vaccine trial with A/Beijing/262/95 in 1998 developed equivalent antibody levels against A/Bayern/ 7/95 and A/Beijing/262/95. Because A/Beijing/262/95-like viruses produce a cross-reactive antibody response to A/Bayern/7/95-like viruses, VRBPAC recommended retaining A/Beijing/262/95 for the 1999-2000 vaccine. Influenza type B isolates from all continents except Asia were similar to B/Beijing/ 184/93, the 1998-99 recommended influenza B vaccine component. In the United States, circulating influenza B viruses remained similar to B/Beijing/184/93. Viruses antigenically related to the B/Victoria/2/87 reference strain were isolated in China, Japan, Singapore, and Thailand and co-circulated with B/Beijing/184/93-like viruses in these countries. However, B/Victoria/2/87-like viruses were not isolated outside of Asia. For the United States, VRBPAC recommended retaining a B/Beijing/184/93-like virus for the vaccine. Manufacturers will use the B/Yamanashi/16/98 strain as the 1999-2000 influenza B vaccine component because of its growth properties and its antigenic similarity to circulating B/Beijing/184/93-like viruses. Reported by: Participating state and territorial epidemiologists and state public health laboratory directors. A Hay, PhD, WHO Collaborating Center for Reference and Research on Influenza, National Institute for Medical Research, London, England. I Gust, MD, A Hampson, WHO Collaborating Center for Reference and Research on Influenza, Parkville, Australia. K Nerome, WHO Collaborating Center for Reference and Research on Influenza, National Institute of Infectious Diseases, Tokyo, Japan. W Lim, FRCPath, Government Virus Unit; M Chan, FHKAM, TA Saw, FHKAM, KH Mak, FHKAM, Dept of Health, Hong Kong, Special Administrative Region, People's Republic of China. L Canas, Armstrong Laboratory, Brooks Air Force Base, Texas. Y Guo, MD, Institute of Virology, National Center for Preventive Medicine, Beijing, People's Republic of China. WHO National Influenza Centers, Div of Emerging and Other Communicable Diseases Surveillance and Control, Geneva, Switzerland. Div of Virology, Center for Biologics Evaluation and Research, Food and Drug Administration. National Respiratory Enteric Virus Surveillance System collaborating laboratories. WHO collaborating laboratories. Sentinel Physicians Influenza Surveillance System. WHO Collaborating Center for Reference and Research on Influenza, Influenza Br, Div of Viral and Rickettsial Diseases, National Center for Infectious Diseases, CDC. Editorial Note:Editorial Note: During the 1998-99 influenza season, both influenza A(H3N2) and influenza B viruses circulated worldwide, and influenza A(H3N2) predominated in the United States. This is the third consecutive year that influenza A(H3N2) viruses have predominated in the United States and the fourth consecutive year in which the proportion of deaths caused by P&I reported by 122 U.S. cities was elevated for several consecutive weeks. Overall, the 1998-99 influenza vaccine strains were well matched with the circulating virus strains. Although influenza epidemics generally peak during December-March each year in temperate regions of the Northern Hemisphere, sporadic cases of influenza and occasionally large outbreaks can occur during the summer (2,3). In temperate regions of the Southern Hemisphere, the influenza season generally peaks during May-August. Influenza epidemics can occur any time of the year in the tropics. Therefore, U.S. physicians should continue to include influenza in the differential diagnosis of febrile respiratory illness during the summer, particularly among travelers to the tropics or Southern Hemisphere or among persons traveling with large international groups. The identification of two cases of human influenza A(H9N2) infection in Hong Kong underscores the need for continued international virologic surveillance for influenza and the timely subtyping of influenza type A isolates. No plans exist to produce a vaccine against influenza A(H9N2). However, several laboratories are working to develop a candidate vaccine should the need arise. Strains to be included in the influenza vaccine usually are selected during the preceding January through March because of scheduling requirements for production, quality control, packaging, distribution, and vaccine administration before the onset of the next influenza season. Recommendations of the Advisory Committee on Immunization Practices for the use of vaccine and antiviral agents for prevention and control of influenza were published in an MMWR Recommendations and Reports on April 30, 1999 (4). References
* Levels of activity are 1) no activity; 2) sporadic--sporadically occurring influenza-like illness (ILI) or culture-confirmed influenza with no outbreaks detected; 3) regional--outbreaks of ILI or culture-confirmed influenza in counties with a combined population of less than 50% of the state's total population; and 4) widespread--outbreaks of ILI or culture-confirmed influenza in counties with a combined population of greater than or equal to 50% of the state's total population. ** An increase of 1.645 standard deviations above the seasonal baseline of P&I deaths is considered the epidemic threshold. Figure 1 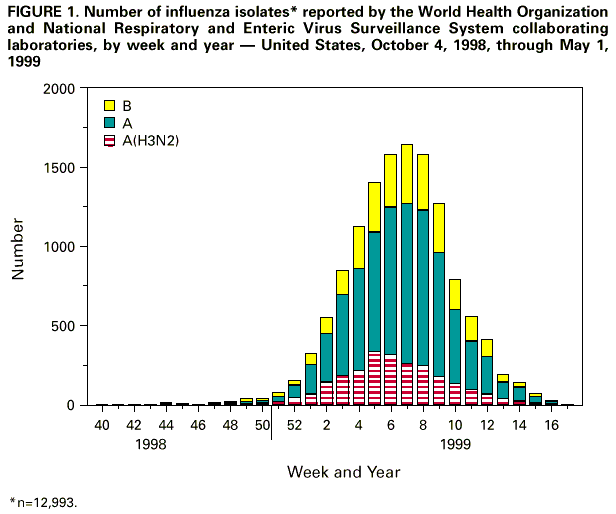 Return to top. Figure 2 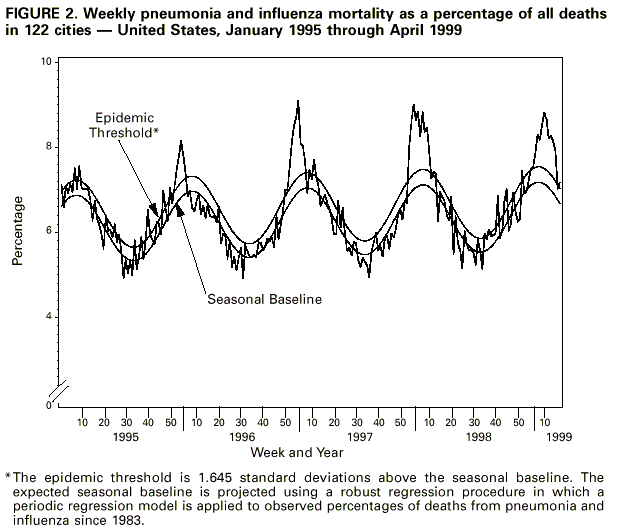 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 5/13/99 |
|||||||||
This page last reviewed 5/2/01
|