 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Update: Outbreak of Influenza A Infection -- Alaska and the Yukon Territory, July-August 1998On July 26, 1998, CDC and Health Canada, in cooperation with local public health authorities, began investigating reports of febrile respiratory illnesses and associated pneumonia among summer land and sea travelers to Alaska and the Yukon Territory (1). Epidemiologic and laboratory evidence has implicated influenza A virus as the etiologic agent of the outbreak. From June 11 through August 22, completed viral cultures of 101 (48%) of 209 nasopharyngeal specimens have yielded 26 influenza A isolates, four other respiratory viruses, and 71 negative results; results are pending for 108 additional specimens. Of the 26 influenza A isolates, five have been characterized at CDC; all have been identified as influenza A/Sydney/5/97 (H3N2)-like viruses, a strain included in the 1998-99 influenza vaccine. This report presents updated information about the outbreak and includes recommendations for influenza A prevention and control in this setting. On August 6, active prospective surveillance was implemented for acute respiratory illnesses (ARI), including influenza-like illness (ILI) and associated pneumonia, in 12 hospitals and clinics in Alaska and the Yukon Territory, a clinic in Seattle and a hospital in Vancouver, and 17 commercial cruise ships touring the region. Because influenza surveillance in North America normally is conducted from October through mid-May, baseline information on the incidence of influenza in Alaska during summer months is not available. As of August 22, prospective surveillance and retrospective case-finding have identified 2199 cases of ARI occurring during May 1-August 22 (Figure_1). Among these illnesses, 766 (35%) cases in tourists and tourism workers in the region met the criteria for ILI (fever or feverishness with cough or sore throat), and an additional 71 (3.2%) cases were confirmed radiographically as pneumonia. Of the persons with pneumonia, 50 required hospitalization. The median age of all persons with ARI was 60 years (range: 1-91 years), and the median age of all persons with pneumonia was 72 years (range: 9-91 years). Since May 1, two deaths have occurred among travelers with ARI to these areas. On May 22, a 79-year-old man developed ILI while on an overland tour and died on June 2. On July 27, a 79-year-old woman developed a respiratory illness the day she completed a 1-week cruise ship tour and died on August 3. Further investigation is under way to determine whether these deaths were associated with influenza A infection. Prospective surveillance continues to identify cases of febrile respiratory illness, particularly among smaller groups of tourists sharing transportation and accommodations on overland tours, and among passengers and crew members on cruise ships. Several cruise lines have initiated active surveillance for respiratory illnesses, organized vaccination campaigns for crew members, and acquired stocks of influenza antiviral medications. As of August 22, active surveillance has identified few (n=46) cases and no outbreaks of influenza among residents in Alaska or the Yukon Territory. Reported by: Special Investigation Team: Div of Public Health, Section of Epidemiology, and Section of Laboratories, Alaska Dept of Health and Social Svcs. Communicable Disease Control, Health and Social Svcs, Yukon Territorial Government; Communicable Disease Epidemiology Svcs and Provincial Laboratory, British Columbia Center for Disease Control Society; Bur of Infectious Diseases, Office of Special Health Initiatives, Field Epidemiology Training Program, and Field Epidemiology Training Program officers, Laboratory Center for Disease Control, Occupational Health and Safety Agency, Health Canada. National Institute for Occupational Safety and Health; National Immunization Program; Div of Applied Public Health Training, Epidemiology Program Office; Arctic Investigations Program, Influenza Br, Div of Viral and Rickettsial Diseases, and Surveillance and Epidemiology Br and Programs Operations Br, Div of Quarantine, National Center for Infectious Diseases; and EIS officers, CDC. Editorial NoteEditorial Note: Each week during May-September, approximately 60,000-70,0000 passengers travel over land and/or by cruise ship to Alaska and the Yukon Territory. Often these travelers use some combination of buses, trains, airplanes, automobiles, and ships for transportation, and many travel as part of large organized groups. Reports of summer outbreaks of influenza A are uncommon, but have been reported previously among groups of travelers in the United States and Canada (2,3). Large organized tour groups often include travelers from various regions of the world, including areas in which seasonal influenza is occurring, which potentially increases the risk for an off-season influenza outbreak among such tourist groups. For example, in August 1997, an influenza outbreak occurred on board a large cruise ship carrying an international group of tourists traveling along the eastern seaboard of the United States and Canada (3). In the influenza outbreak described in this report, cases of respiratory illness have occurred among tourists during different tours managed by different companies. Preliminary evidence suggests that most illnesses appear to have developed during land-based travel. However, cases of illness consistent with influenza transmission on board cruise ships are being reported in passengers and crew members. ARI and ILI cases that occurred before active surveillance was implemented probably have been substantially underreported. In response to the outbreak, CDC and Health Canada jointly used the following considerations to develop recommendations for travelers to the region: 1) new cases of ILI continue to occur among tourists in the region, 2) the tourist season will wane substantially during the next 2-3 weeks, and 3) influenza vaccine availability at this time of year is limited. No special prevention measures are recommended for travelers who are aged less than 65 years and in good health. These travelers are unlikely to develop a febrile respiratory illness during their travels, and the risk for serious complications from influenza is low in this group. Early general vaccination of the resident populations in Alaska and the Yukon Territory also is not recommended at this time. On the basis of these considerations, persons who are aged greater than or equal to 65 years or who have certain underlying chronic medical conditions (e.g., pulmonary or cardiac disease) are recommended to consult their physicians before traveling to Alaska and the Yukon Territory this summer. This group is at increased risk for serious complications from influenza, including pneumonia, hospitalization, and death (4). These persons should receive information about the signs and symptoms of influenza and about the advisability of carrying rimantadine or amantadine, antiviral medications that can be used for the treatment or prophylaxis of influenza A infections (but not influenza B). Both antiviral medications can reduce the duration of influenza A illness and viral shedding if administered within 48 hours of onset of symptoms, but also may lead to central nervous system or gastrointestinal side effects and may require dosage adjustments in patients with underlying renal or hepatic disease. Physicians and other health-care providers in Alaska, in the Yukon Territory, and on board cruise ships who may provide care for persons with ILI should consider obtaining commercially available rapid antigen-detection kits for influenza and supplies of rimantadine or amantadine for the treatment of patients with febrile respiratory illness. All cruise lines and overland tour companies should implement active surveillance for ARI among travelers and employees for the remainder of the Alaska/Yukon tour season, which ends October 1, 1998. Tour companies that conduct combined land and sea tours in the region should offer vaccination with 1998-99 influenza vaccine to all of their staff who have direct contact with high-risk travelers in this area. Although vaccination of staff during the final 2 weeks of the Alaska tour season is unlikely to have a large effect in limiting the scope of disease in travelers during this outbreak, it will help to decrease the transmission of influenza to new groups of passengers after the ships depart Alaskan waters for other regions of the world. Health-care providers evaluating patients with febrile respiratory illnesses or pneumonia should obtain a travel history and consider influenza A in the differential diagnosis for persons who have traveled recently to Alaska or the Yukon Territory. Information about the outbreak is available on the CDC World-Wide Web site, http://www.cdc.gov/travel/travel.html. Health-care providers may continue to report cases of illness to the Special Investigation Team, telephone (907) 729-3431, fax (907) 729-3429, or e-mail SITEAM@cdc.gov. References
Figure_1 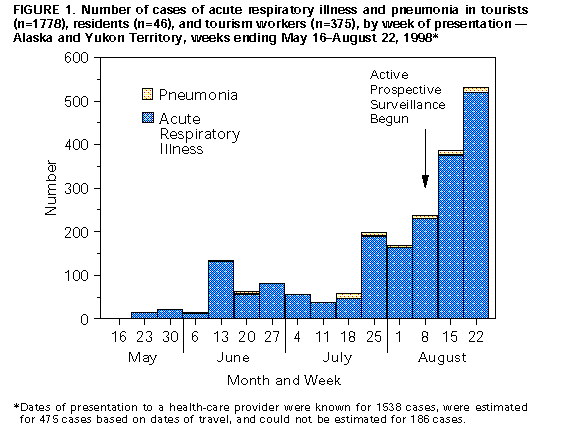 Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 10/05/98 |
|||||||||
This page last reviewed 5/2/01
|