This page is a historical archive and is no longer maintained.
For current information, please visit http://www.cdc.gov/media/
Press Release
For Immediate Release: Thursday, December 12, 2013
Contact:
Media Relations
(404) 639-3286
Public gets early snapshot of MRSA and C. difficile infections in individual hospitals
CDC, CMS collaborate to advance public reporting of important hospital quality indicators
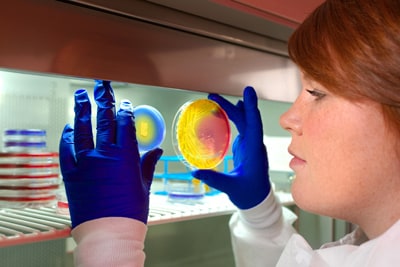
CDC microbiologist, Valerie Albrecht, holds up two plates of methicillin-resistant Staphylococcus aureus (MRSA).
New data posted today and gathered through the Centers for Disease Control and Prevention's (CDC) National Healthcare Safety Network (NHSN) gives patients a first look at how their local hospitals are doing at preventing Clostridium difficile infections (deadly diarrhea) and methicillin-resistant Staphylococcus aureus (MRSA) bloodstream infections. This information, as well as other hospital performance measures, is collected as part of the Centers for Medicare & Medicaid Services' (CMS) Hospital Inpatient Quality Reporting (IQR) Program and is publicly available on the Hospital Compare website.
The numbers represent only the first quarter of 2013; measurements of how hospitals are doing will be more precise and provide a more complete picture as more information is collected over time. The next update, which will represent six months of data, is scheduled for April 2014.
"Sunlight is a great disinfectant, and public reporting of hospital infections is the sunlight the public has asked for and deserves when it comes to their health and safety," said CDC Director Tom Frieden, M.D., M.P.H. "Hospitals understand the importance of reporting, and their leaders are using this information to prevent infections and keep their patients safer."
The Hospital IQR Program uses a variety of tools to help stimulate and support improvements in patient care, including the Hospital Compare website, which helps distribute objective, easy-to-understand data on hospital performance.
"The Hospital Compare website enables consumers to make informed choices and gives hospital leaders and their staff comparative information to help drive improvement," said Patrick Conway, M.D., CMS chief medical officer and director of the Center for Clinical Standards and Quality. "Central line bloodstream infections have decreased more than 40 percent through transparency and improvement efforts, which has saved thousands of lives, and we hope to see the same positive results for these two common infections."
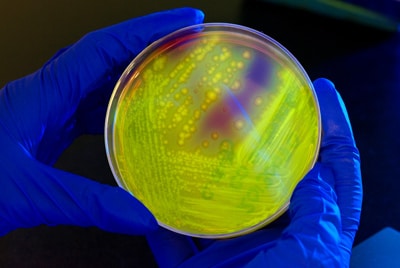
Yellow-green fluorescence of Clostridium difficile under long-wave UV irradiation on a CCFA plate.
C. difficile causes at least 250,000 hospitalizations and 14,000 deaths every year, and was recently categorized by CDC as an urgent threat to patient safety. On the other hand, although still a common and severe threat to patients, invasive MRSA infections in healthcare settings appear to be declining. Between 2005 and 2011 overall rates of invasive MRSA dropped 31 percent. Success began with preventing central-line-associated bloodstream infections caused by MRSA, for which rates fell nearly 50 percent from 1997 to 2007.
Some facilities that do not currently have a sufficient amount of data to collect may not have their infection ratios included in the Hospital IQR Program and subsequently, on the Hospital Compare website. For example, the number of C. difficile and MRSA bloodstream infections in some smaller facilities might not provide enough information to calculate infection ratios until they report additional calendar quarters of data.
In accordance with the clinical quality measure used by CMS and CDC for laboratory-identified C. difficile and MRSA bloodstream infections, the Hospital Compare website only reflects hospital-onset infections, which are defined as those detected after patients are hospitalized for a minimum of three days. Patients whose infections arose outside of the hospital are not included in the infection counts for the quality measure.
Major teaching hospitals, hospitals with more than 400 beds and those with high community-onset rates continue to have the highest risk for C. difficile and MRSA bloodstream infections, all of which is taken into account by risk adjustment when the clinical quality measure is calculated.
CDC and CMS continue to provide hospitals with the training and tools to look deeper into their healthcare-associated infection data to target prevention efforts. Hospitals are encouraged to participate in a variety of federal healthcare-associated infection prevention efforts, including those made available through state health departments, CMS Quality Improvement Organizations, and Partnership for Patients Hospital Engagement Networks.
Additionally, CDC is working with hospitals and healthcare information technology vendors to build capacity for electronic reporting of antibiotic use and resistance data to NHSN. This work includes combining data from hospital admission, discharge, and patient transfer information (ADT) systems with data from medication administration and laboratory information systems. More systematic reporting of antibiotic use and resistance data will help hospitals assure that powerful medications are used appropriately. Additional data from ADT systems, including transfer destinations also will provide a comprehensive view of patient movement between facilities and readmissions associated with MRSA and C. difficile infections. Given that these infections can arise in non-hospital settings, tracking patient movements and the onset of infections is critical to protecting patients and saving lives.
Get email updates
To receive email updates about this page, enter your email address:
Contact Us:
- Centers for Disease Control and Prevention
1600 Clifton Rd
Atlanta, GA 30333 - 800-CDC-INFO
(800-232-4636)
TTY: (888) 232-6348 - Contact CDC–INFO
 ShareCompartir
ShareCompartir


