Weekly U.S. Influenza Surveillance Report
Updated April 12, 2024

Seasonal influenza activity remains elevated nationally but continues to decrease.
Viruses
All data are preliminary and may change as more reports are received.
Directional arrows indicate changes between the current week and the previous week. Additional information on the arrows can be found at the bottom of this page.
A description of the CDC influenza surveillance system, including methodology and detailed descriptions of each data component is available on the surveillance methods page.
Additional information on the current and previous influenza seasons for each surveillance component are available on FluView Interactive.
Key Points
- Seasonal influenza activity remains elevated but continues to decrease nationally.
- Nationally, percent positivity for both influenza A and B decreased compared to last week.
- Nationally, outpatient respiratory illness declined and is below baseline for the first time since late October.1 Regions 2, 3, 4, 6, 8, 9, and 10 are below their baselines, while HHS regions 1, 5 and 7 remain above their region-specific baselines.
- Nationally, the number of weekly flu hospital admissions has been decreasing since January.
- During Week 14, of the 272 viruses reported by public health laboratories, 160 (58.8%) were influenza A and 112 (41.2%) were influenza B. Of the 104 influenza A viruses subtyped during Week 14, 45 (43.3%) were influenza A(H1N1)pdm09 and 59 (56.7%) were A(H3N2).
- Five influenza-associated pediatric deaths occurring during the 2023-2024 season were reported to CDC during Week 14, bringing the season total to 138 pediatric deaths.
- CDC estimates that there have been at least 33 million illnesses, 360,000 hospitalizations, and 23,000 deaths from flu so far this season.
- CDC recommends that everyone 6 months and older get an annual flu vaccine as long as influenza viruses are spreading.2 Vaccination can still provide benefit this season.
- There also are prescription flu antiviral drugs that can treat flu illness; those should be started as early as possible and are especially important for higher risk patients.3
- Flu viruses are among several viruses contributing to respiratory disease activity. CDC is providing updated, integrated information about COVID-19, flu, and RSV activity on a weekly basis.
U.S. Virologic Surveillance
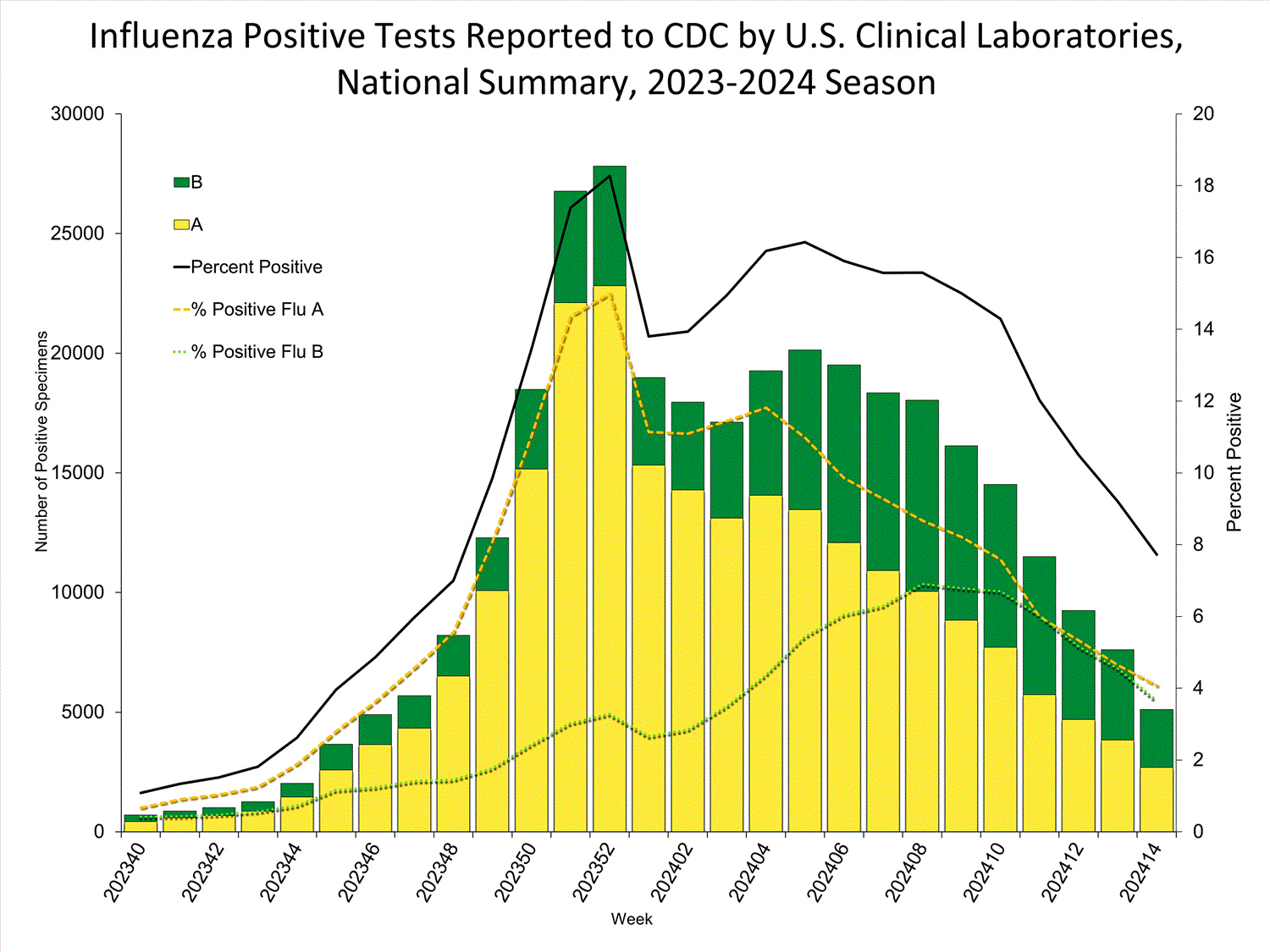
Nationally, the percentage of respiratory specimens testing positive for influenza in clinical laboratories decreased (change of >0.5 percentage points) compared to the previous week. Regions 1, 2, 3, 5, 6, 8, and 10 decreased in percent positivity; regions 4 and 9 remained stable, and Region 7 reported a slight increase during Week 14 compared to Week 13. The regions with the highest percent positivity were regions 7 (17.6%), 5 (11.2%), and 1 (8.7%). Nationally, influenza A(H1N1)pdm09, A(H3N2), and B/Victoria viruses are all co-circulating. However, the distribution of circulating viruses varies by region. For regional and state level data and age group distribution, please visit FluView Interactive. Viruses known to be associated with recent receipt of live attenuated influenza vaccine (LAIV) or found upon further testing to be a vaccine virus are not included, as they are not circulating influenza viruses.
Clinical Laboratories
The results of tests performed by clinical laboratories nationwide are summarized below. Data from clinical laboratories (the percentage of specimens tested that are positive for influenza virus) are used to monitor whether influenza activity is increasing or decreasing.
| Week 14 |
Data Cumulative since October 1, 2023 (Week 40) |
|
|---|---|---|
| No. of specimens tested | 66,199 | 2,837,222 |
| No. of positive specimens (%) | 5,111 (7.7%) | 327,063 (11.5%) |
| Positive specimens by type | ||
| Influenza A | 2,692 (52.7%) | 228,076 (69.7%) |
| Influenza B | 2,419 (47.3%) | 98,977 (30.3%) |
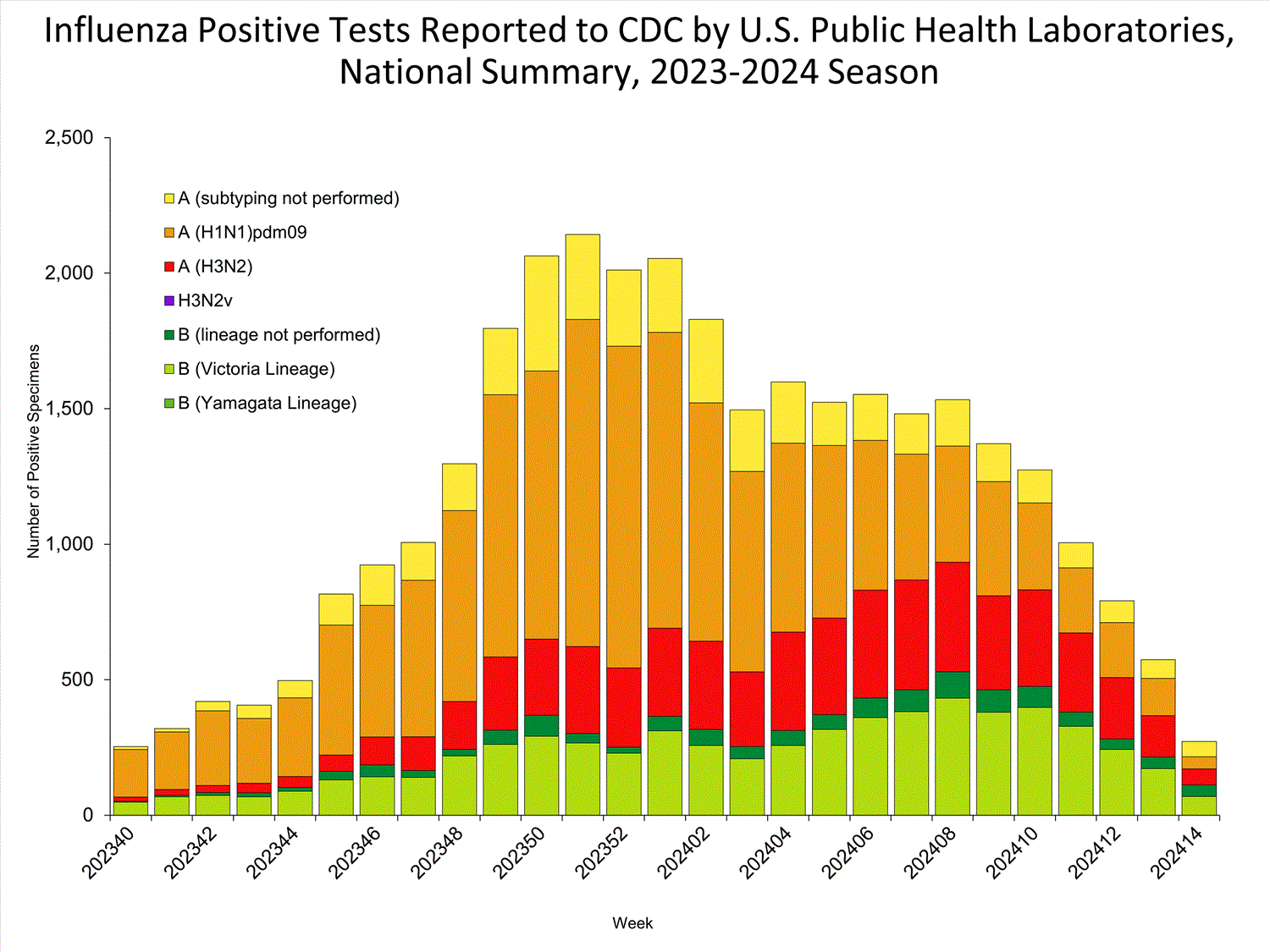
Public Health Laboratories
The results of tests performed by public health laboratories nationwide are summarized below. Data from public health laboratories are used to monitor the proportion of circulating influenza viruses that belong to each influenza subtype/lineage.
| Week 14 |
Data Cumulative since October 1, 2023 (Week 40) |
|
|---|---|---|
| No. of specimens tested | 1,117 | 98,281 |
| No. of positive specimens | 272 | 32,302 |
| Positive specimens by type/subtype | ||
| Influenza A | 160 (58.8%) | 24,958 (77.3%) |
| Subtyping Performed | 104 (65.0%) | 20,711 (83.0%) |
| (H1N1)pdm09 | 45 (43.3%) | 14,652 (70.7%) |
| H3N2 | 59 (56.7%) | 6,059 (29.3%) |
| H3N2v | 0 (0.0%) | 0 (0.0%) |
| Subtyping not performed | 56 (35.0%) | 4,247 (17.0%) |
| Influenza B | 112 (41.2%) | 7,344 (22.7%) |
| Lineage testing performed | 69 (61.6%) | 6,140 (83.6%) |
| Yamagata lineage | 0 (0.0%) | 0 (0.0%) |
| Victoria lineage | 69 (100.0%) | 6,140 (100.0%) |
| Lineage not performed | 43 (38.4%) | 1,204 (16.4%) |
Additional virologic surveillance information for current and past seasons:
Surveillance Methods | FluView Interactive: National, Regional, and State Data or Age Data
Influenza Virus Characterization
CDC performs genetic and antigenic characterization of U.S. viruses submitted from state and local public health laboratories according to the Right Size Roadmap submission guidance. These data are used to compare how similar the currently circulating influenza viruses are to the reference viruses representing viruses contained in the current influenza vaccines. The data are also used to monitor evolutionary changes that continually occur in influenza viruses circulating in humans. CDC also tests susceptibility of circulating influenza viruses to antiviral medications including the neuraminidase inhibitors (oseltamivir, zanamivir, and peramivir) and the PA endonuclease inhibitor baloxavir.
CDC has genetically characterized 3,369 influenza viruses collected since October 1, 2023.
| Virus Subtype or Lineage | Genetic Characterization | ||||
|---|---|---|---|---|---|
| Total No. of Subtype/Lineage Tested |
HA Clade |
Number (% of subtype/lineage tested) |
HA Subclade |
Number (% of subtype/lineage tested) |
|
| A/H1 | 1,378 | ||||
| 6B.1A.5a | 1,378 (100%) | 2a | 339 (24.6%) | ||
| 2a.1 | 1,039 (75.4%) | ||||
| A/H3 | 1,068 | ||||
| 3C.2a1b.2a | 1,068 (100%) | 2a.1b | 1 (0.1%) | ||
| 2a.3a | 1 (0.1%) | ||||
| 2a.3a.1 | 1,065 (99.7%) | ||||
| 2b | 1 (0.1%) | ||||
| B/Victoria | 923 | ||||
| V1A | 923 (100%) | 3a.2 | 923 (100%) | ||
| B/Yamagata | 0 | ||||
| Y3 | 0 | Y3 | 0 (0%) | ||
CDC antigenically characterizes influenza viruses by hemagglutination inhibition (HI) (H1N1pdm09, H3N2, B/Victoria, and B/Yamagata viruses) or neutralization-based HINT (H3N2 viruses) using antisera that ferrets make after being infected with reference viruses representing the 2023-2024 Northern Hemisphere recommended cell or recombinant-based vaccine viruses. Antigenic differences between viruses are determined by comparing how well the antibodies made against the vaccine reference viruses recognize the circulating viruses that have been grown in cell culture. Ferret antisera are useful because antibodies raised against a particular virus can often recognize small changes in the surface proteins of other viruses. In HI assays, viruses with similar antigenic properties have antibody titer differences of less than or equal to 4-fold when compared to the reference (vaccine) virus. In HINT, viruses with similar antigenic properties have antibody neutralization titer differences of less than or equal to 8-fold. Viruses selected for antigenic characterization are a subset representing the genetic changes in the surface proteins seen in genetically characterized viruses.
Influenza A Viruses
- A (H1N1)pdm09: 217 A(H1N1)pdm09 viruses were antigenically characterized by HI, and all were well-recognized (reacting at titers that were within 4-fold of the homologous virus titer) by ferret antisera to cell-grown A/Wisconsin/67/2022-like reference viruses representing the A(H1N1)pdm09 component for the cell- and recombinant-based influenza vaccines.
- A (H3N2): 249 A(H3N2) viruses were antigenically characterized by HI or HINT, and 246 (98.8%) were well-recognized (reacting at titers that were within 4-fold of the homologous virus titer in HI or reacting at titers that were less than or equal to 8-fold of the homologous virus in HINT) by ferret antisera to cell-grown A/Darwin/6/2021-like reference viruses representing the A(H3N2) component for the cell- and recombinant-based influenza vaccines.
Influenza B Viruses
- B/Victoria: 213 influenza B/Victoria-lineage virus were antigenically characterized by HI, and all were well-recognized (reacting at titers that were within 4-fold of the homologous virus titer) by ferret antisera to cell-grown B/Austria/1359417/2021-like reference viruses representing the B/Victoria component for the cell- and recombinant-based influenza vaccines.
- B/Yamagata: No influenza B/Yamagata-lineage viruses were available for antigenic characterization.
Assessment of Virus Susceptibility to Antiviral Medications
CDC assesses susceptibility of influenza viruses to the antiviral medications including the neuraminidase inhibitors (oseltamivir, zanamivir, and peramivir) and the PA endonuclease inhibitor baloxavir using next generation sequence analysis supplemented by laboratory assays. Information about antiviral susceptibility test methods can be found at U.S. Influenza Surveillance: Purpose and Methods | CDC.
Viruses collected in the U.S. since October 01, 2023, were tested for antiviral susceptibility as follows:
| Antiviral Medication | Total Viruses | A/H1 | A/H3 | B/Victoria | ||
|---|---|---|---|---|---|---|
| Neuraminidase Inhibitors | Oseltamivir | Viruses Tested | 3,350 | 1,379 | 1,062 | 909 |
| Reduced Inhibition | 1 (0.03%) | 1 (0.07%) | 0 (0.0%) | 0 (0.0%) | ||
| Highly Reduced Inhibition | 2 (0.06%) | 2 (0.1%) | 0 (0.0%) | 0 (0.0%) | ||
| Peramivir | Viruses Tested | 3,350 | 1,379 | 1,062 | 909 | |
| Reduced Inhibition | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | ||
| Highly Reduced Inhibition | 2 (0.06%) | 2 (0.1%) | 0 (0.0%) | 0 (0.0%) | ||
| Zanamivir | Viruses Tested | 3,350 | 1,379 | 1,062 | 909 | |
| Reduced Inhibition | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | ||
| Highly Reduced Inhibition | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | 0 (0.0%) | ||
| PA Cap-Dependent Endonuclease Inhibitor | Baloxavir | Viruses Tested | 3,261 | 1,340 | 1,039 | 882 |
| Decreased Susceptibility | 1 (0.03%) | 0 (0.0%) | 1 (0.1%) | 0 (0.0%) | ||
Two A(H1N1)pdm09 viruses had NA-H275Y amino acid substitution and showed highly reduced inhibition by oseltamivir and peramivir. One (H1N1)pdm09 virus had NA-I223V and NA-S247N amino acid substitutions and showed reduced inhibition by oseltamivir. One A(H3N2) virus had PA-I38T amino acid substitution and showed reduced susceptibility to baloxavir.
High levels of resistance to the adamantanes (amantadine and rimantadine) persist among influenza A(H1N1)pdm09 and influenza A(H3N2) viruses (the adamantanes are not effective against influenza B viruses). Therefore, use of these antivirals for treatment and prevention of influenza A virus infection is not recommended and data from adamantane resistance testing are not presented.
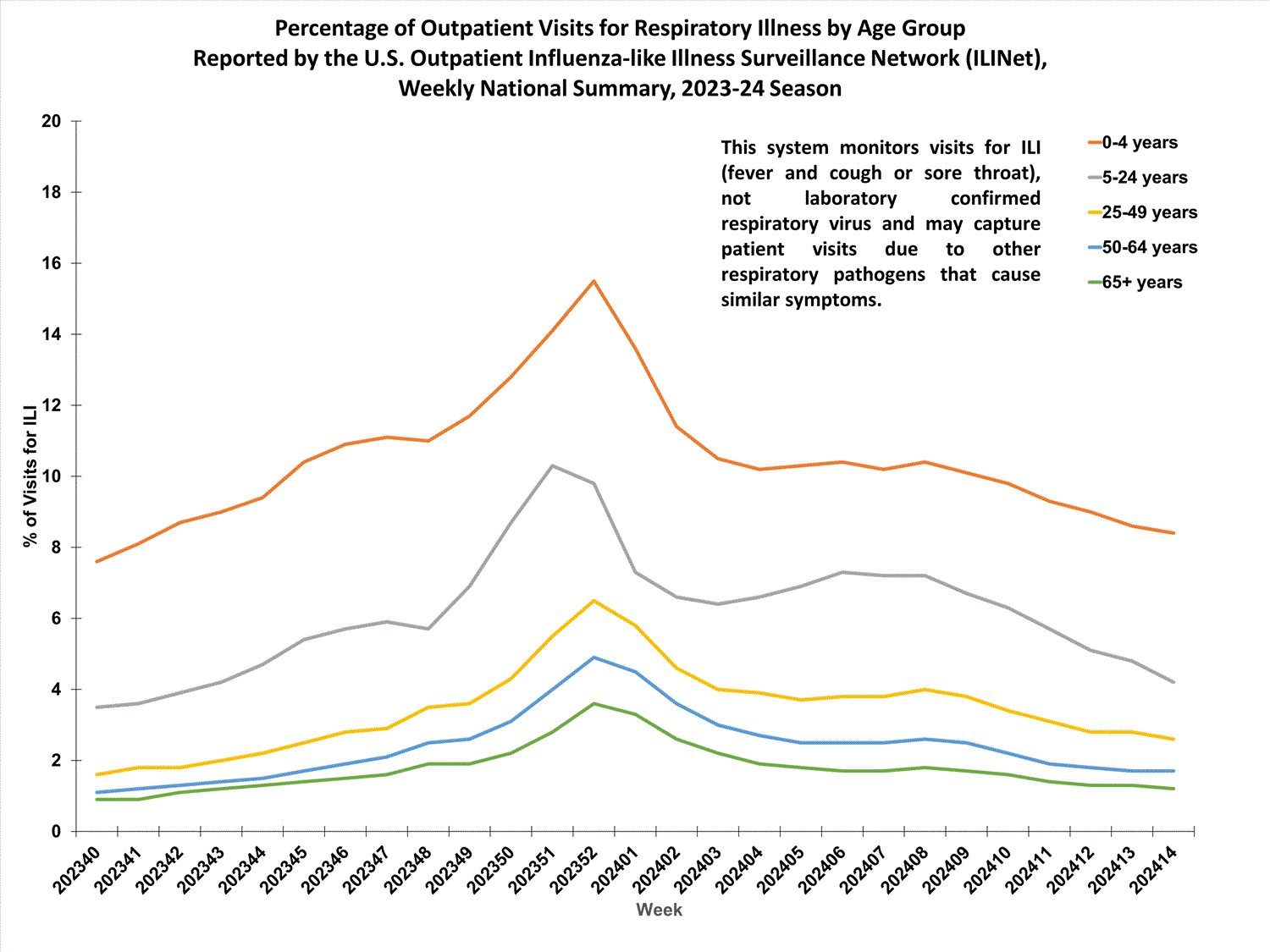
Outpatient Respiratory Illness Surveillance
The U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet) monitors outpatient visits for respiratory illness referred to as influenza-like illness [ILI (fever plus cough or sore throat)], not laboratory-confirmed influenza and will therefore capture respiratory illness visits due to infection with pathogens that can present with similar symptoms, including influenza viruses, SARS-CoV-2, and RSV. It is important to evaluate syndromic surveillance data, including that from ILINet, in the context of other sources of surveillance data to obtain a more complete and accurate picture of influenza, SARS-CoV-2, and other respiratory virus activity. CDC is providing integrated information about COVID-19, influenza, and RSV activity on a website that is updated weekly. Information about other respiratory virus activity can be found on CDC’s National Respiratory and Enteric Virus Surveillance System (NREVSS) website.
Outpatient Respiratory Illness Visits
Nationally, during Week 14, 2.8% of patient visits reported through ILINet were due to respiratory illness that included fever plus a cough or sore throat, also referred to as ILI. This has decreased (change of ≥ 0.1 percentage points) since Week 13 and is now below the national baseline. The percentage of visits for ILI decreased in regions 2, 4, 5, 6, 7, and 8, and remained stable in all other regions in Week 14 compared to Week 13. Regions 1, 5, and 7 are above their baseline, and all other regions are below their respective baselines. Multiple respiratory viruses are co-circulating, and the relative contribution of influenza virus infection to ILI varies by location.
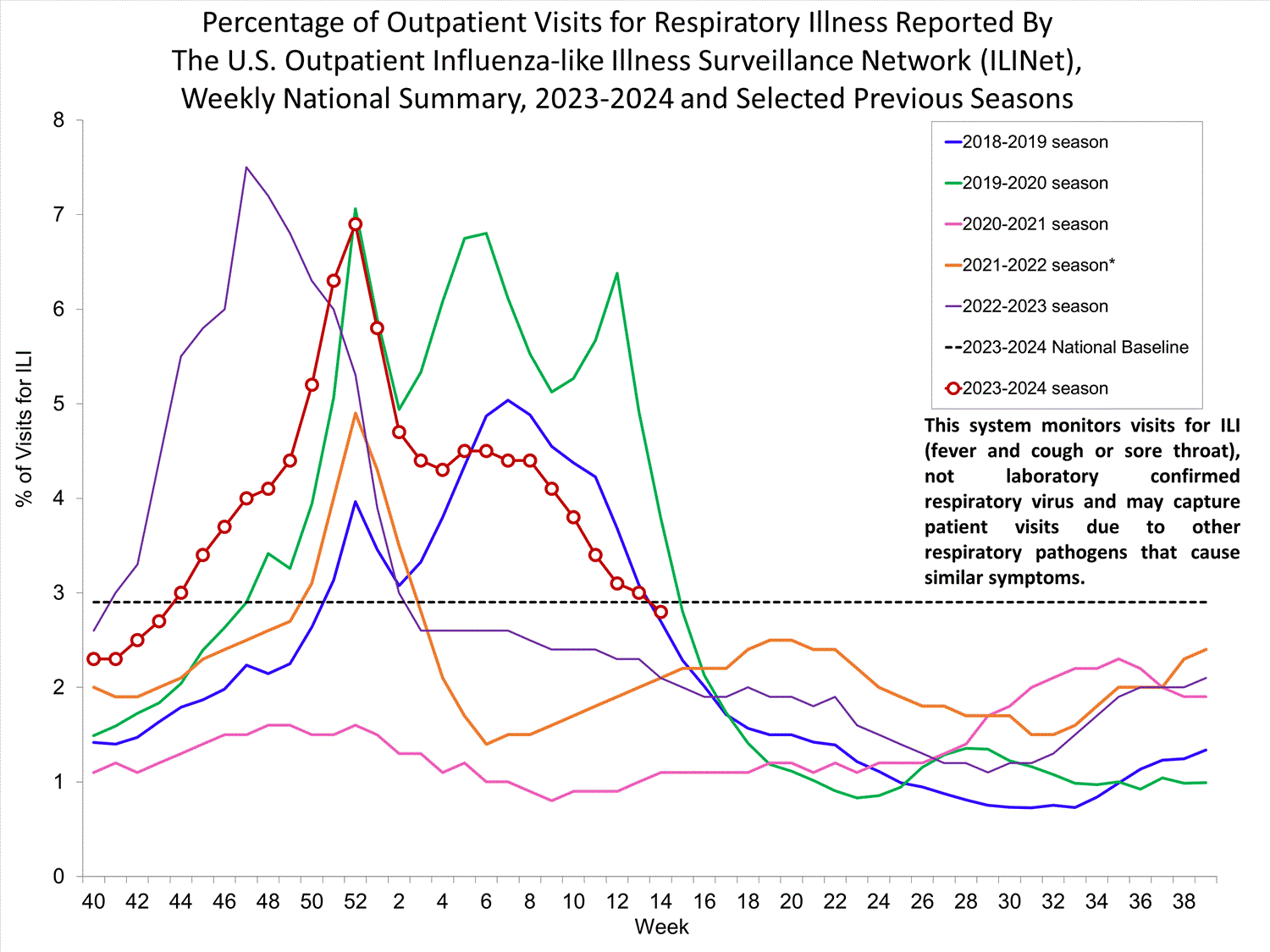
* Effective October 3, 2021 (week 40), the ILI definition (fever plus cough or sore throat) no longer includes “without a known cause other than influenza.”
Outpatient Respiratory Illness Visits by Age Group
About 70% of ILINet participants provide both the number of patient visits for respiratory illness and the total number of patient visits for the week broken out by age group. Data from this subset of providers are used to calculate the percentages of patient visits for respiratory illness by age group.
The percentage of visits for respiratory illness reported in ILINet decreased in the 0-4 years, 5-24 years, and 25-49 years age groups, and remained stable in the 50-64 years and 65+ years age groups in Week 14 compared to Week 13.
Outpatient Respiratory Illness Activity Map
Data collected in ILINet are used to produce a measure of ILI activity* by state/jurisdiction and Core Based Statistical Areas (CBSA).
| Activity Level | Number of Jurisdictions | Number of CBSAs | ||
|---|---|---|---|---|
| Week 14 (Week ending Apr. 6, 2024) |
Week 13 (Week ending Mar. 30, 2024) |
Week 14 (Week ending Apr. 6, 2024) |
Week 13 (Week ending Mar. 30, 2024) |
|
| Very High | 0 | 0 | 0 | 1 |
| High | 1 | 6 | 13 | 20 |
| Moderate | 7 | 5 | 37 | 53 |
| Low | 13 | 21 | 127 | 136 |
| Minimal | 33 | 23 | 513 | 495 |
| Insufficient Data | 1 | 0 | 239 | 224 |
*Data collected in ILINet may disproportionally represent certain populations within a jurisdiction or CBSA, and therefore, may not accurately depict the full picture of influenza activity for the entire jurisdiction or CBSA. Differences in the data presented here by CDC and independently by some health departments likely represent differing levels of data completeness with data presented by the health department likely being the more complete.
Additional information about medically attended visits for ILI for current and past seasons:
Surveillance Methods | FluView Interactive: National, Regional, and State Data or ILI Activity Map
Hospitalization Surveillance
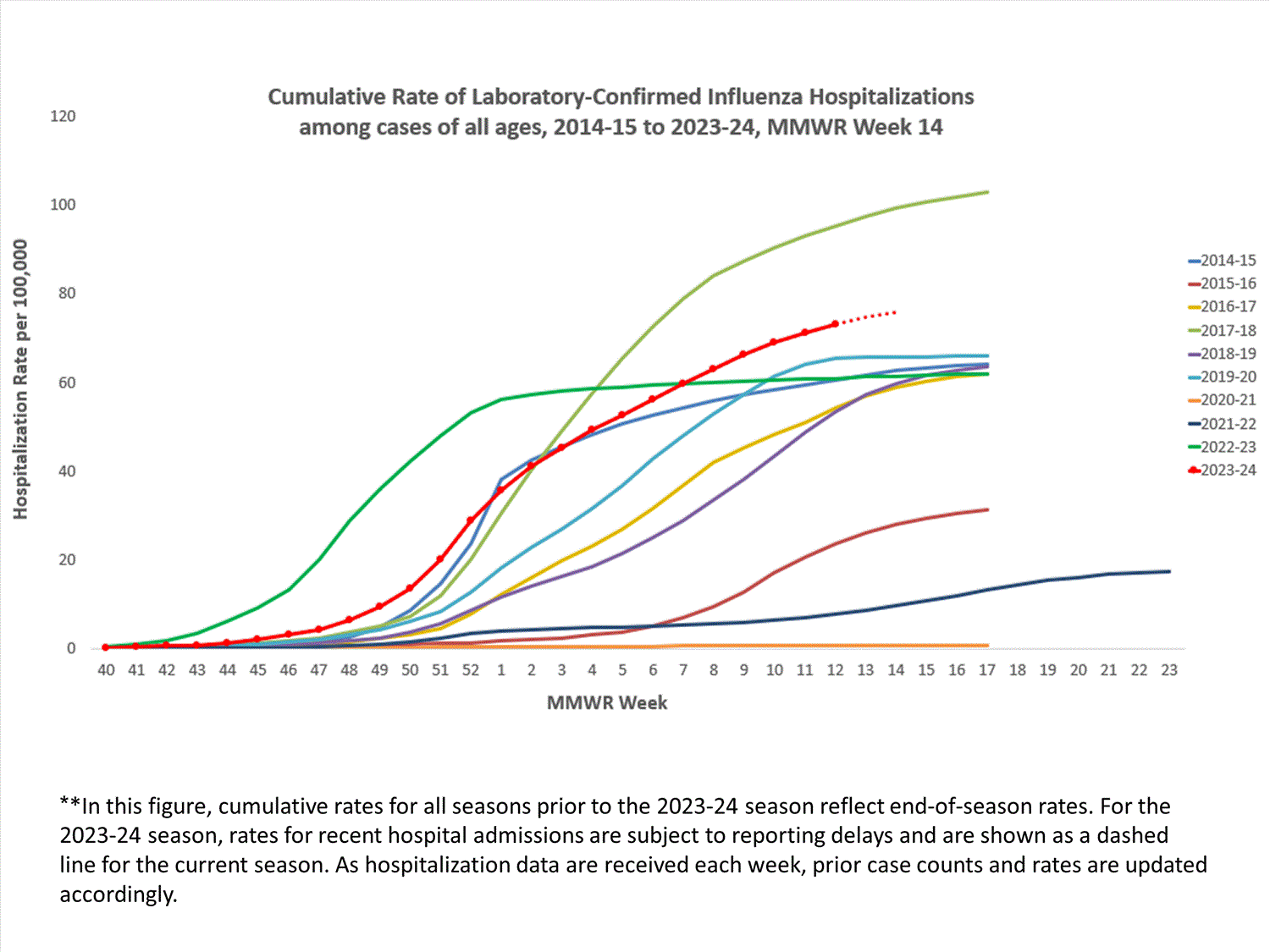
FluSurv-NET
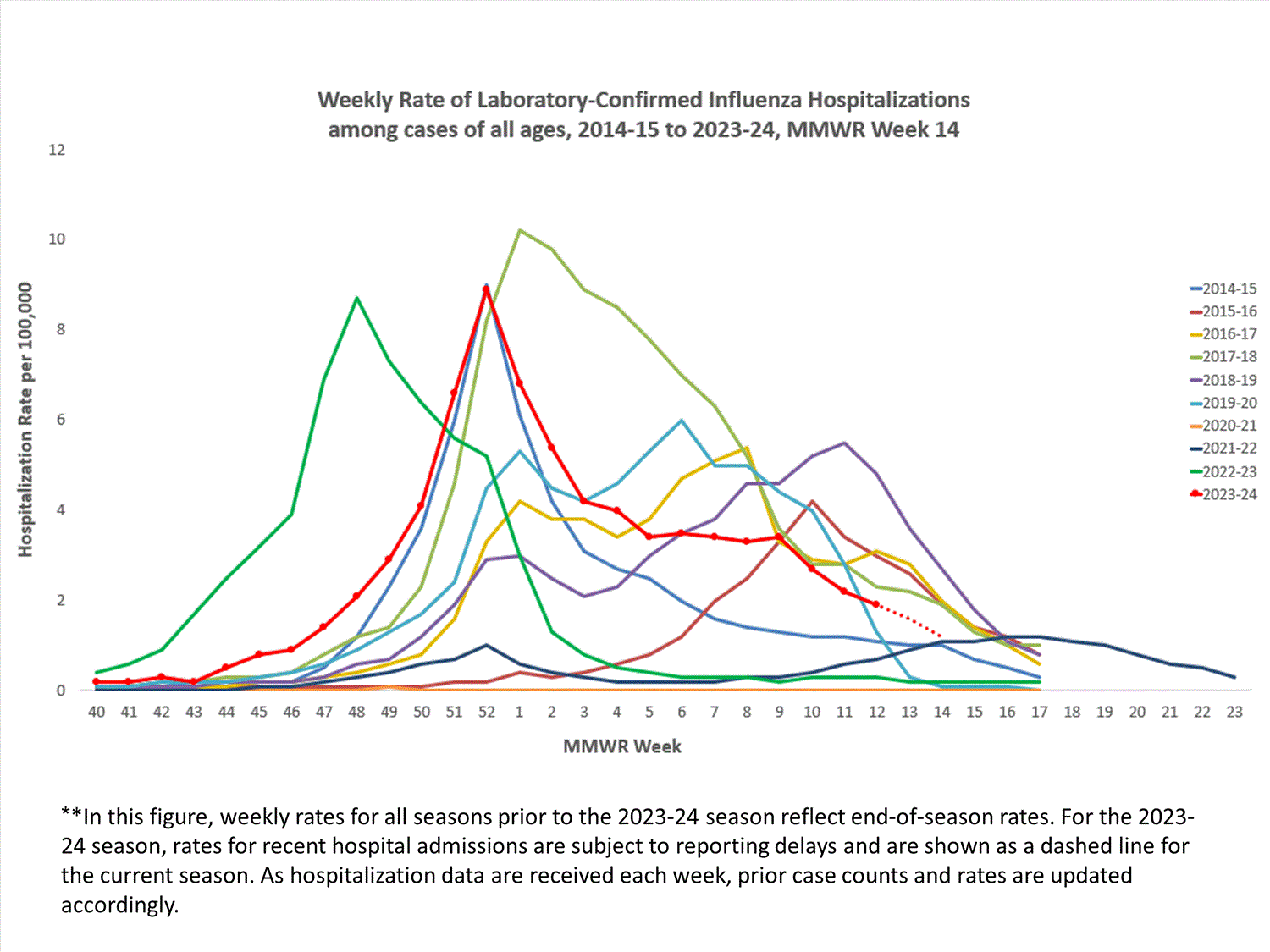
The Influenza Hospitalization Surveillance Network (FluSurv-NET) conducts population-based surveillance for laboratory-confirmed influenza-related hospitalizations in select counties in 14 states and represents approximately 9% of the U.S. population. FluSurv-NET hospitalization data are preliminary. As data are received each week, prior case counts and rates are updated accordingly.
A total of 23,235 laboratory-confirmed influenza-associated hospitalizations were reported by FluSurv-NET sites between October 1, 2023, and April 6, 2024. The weekly hospitalization rate observed in Week 14 was 1.2 per 100,000 population. The weekly hospitalization rate observed during Week 52 is the third highest peak weekly rate observed during all seasons going back to 2010-2011 following the 2014-2015 and 2017-2018 seasons. The overall cumulative hospitalization rate was 76.0 per 100,000 population. This cumulative hospitalization rate is the second highest cumulative hospitalization rate when compared against previous end-of-season rates for Week 14, and it is the second highest cumulative in-season hospitalization rate observed in Week 14, following the 2017-2018 season (101.6). Cumulative in-season hospitalization rates observed in Week 14 from 2010-2011 through 2022-2023 ranged from 0.8 to 68.2.
When examining rates by age, the highest cumulative hospitalization rate per 100,000 population was among adults aged 65 years and older (207.5), followed by adults aged 50-64 years (90.2) and children aged 0-4 years (78.0). When examining age-adjusted rates by race and ethnicity, the highest rate of hospitalization per 100,000 population was among non-Hispanic Black persons (139.8), followed by non-Hispanic American Indian or Alaska Native persons (104.8), Hispanic persons (70.8), non-Hispanic White persons (58.5), and non-Hispanic Asian/Pacific Islander persons (39.8).
Among 23,235 hospitalizations, 19,767 (85.1%) were associated with influenza A virus, 3,307 (14.2%) with influenza B virus, 42 (0.2%) with influenza A virus and influenza B virus co-infection, and 118 (0.5%) with influenza virus for which the type was not determined. Among those with influenza A subtype information, 3,319 (71.0%) were A(H1N1) pdm09 and 1,358 (29.0%) were A(H3N2).
Among 2,879 hospitalized adults with information on underlying medical conditions, 95.4% had at least one reported underlying medical condition, the most commonly reported were hypertension, cardiovascular disease, obesity, and metabolic disease. Among 1,381 hospitalized women of childbearing age (15-49 years) with information on pregnancy status, 22.1% were pregnant. Among 898 hospitalized children with information on underlying medical conditions, 69.5% had at least one reported underlying medical condition; the most commonly reported was asthma, followed by obesity and neurologic disease.
In these figures, cumulative and weekly rates for all seasons prior to the 2023-2024 season reflect end-of-season rates. For the 2023-2024 season, rates for recent hospitals admissions are subject to reporting delays. As hospitalization data are reviewed each week, prior case counts and rates are updated accordingly.
Additional FluSurv-NET hospitalization surveillance information for current and past seasons and additional age groups:
Surveillance Methods |FluView Interactive: Rates by Age, Sex, and Race/Ethnicity or Data on Patient Characteristics | RESP-NET Interactive
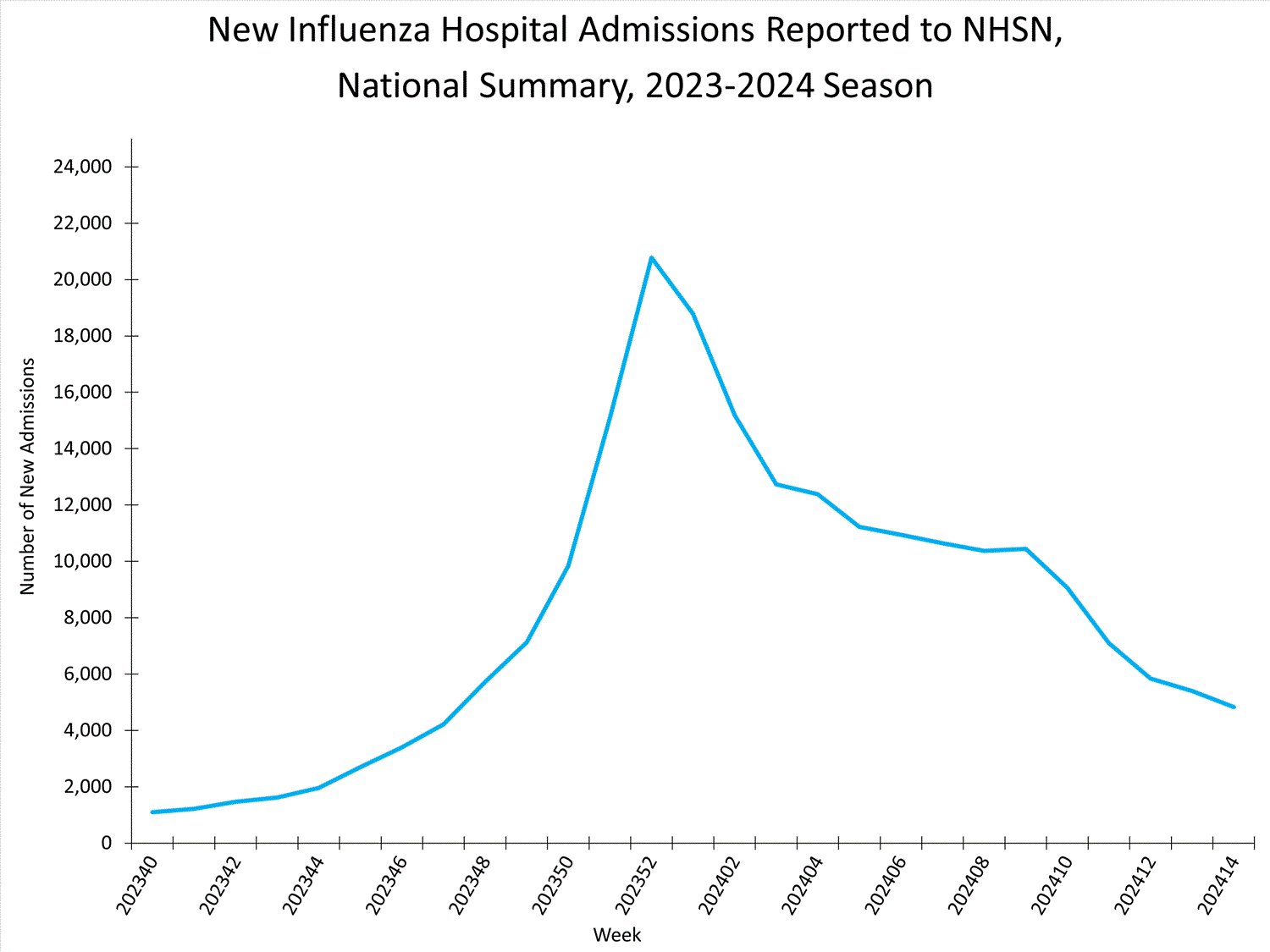
National Healthcare Safety Network (NHSN) Hospitalization Surveillance
Hospitals report to NHSN the weekly number of patients admitted with laboratory-confirmed influenza. During Week 14, 4,825 patients with laboratory-confirmed influenza were admitted to a hospital. The number of patients admitted to a hospital with laboratory-confirmed influenza for Week 14 decreased (change of >5%) compared to Week 13 nationally and in regions 4, 5, 6, 7, 8, 9, and 10. The number of hospitalizations remained stable in regions 1 and 2 and slightly increased in Region 3.
Additional NHSN Hospitalization Surveillance information:
Surveillance Methods | Additional Data | FluView Interactive
Mortality Surveillance
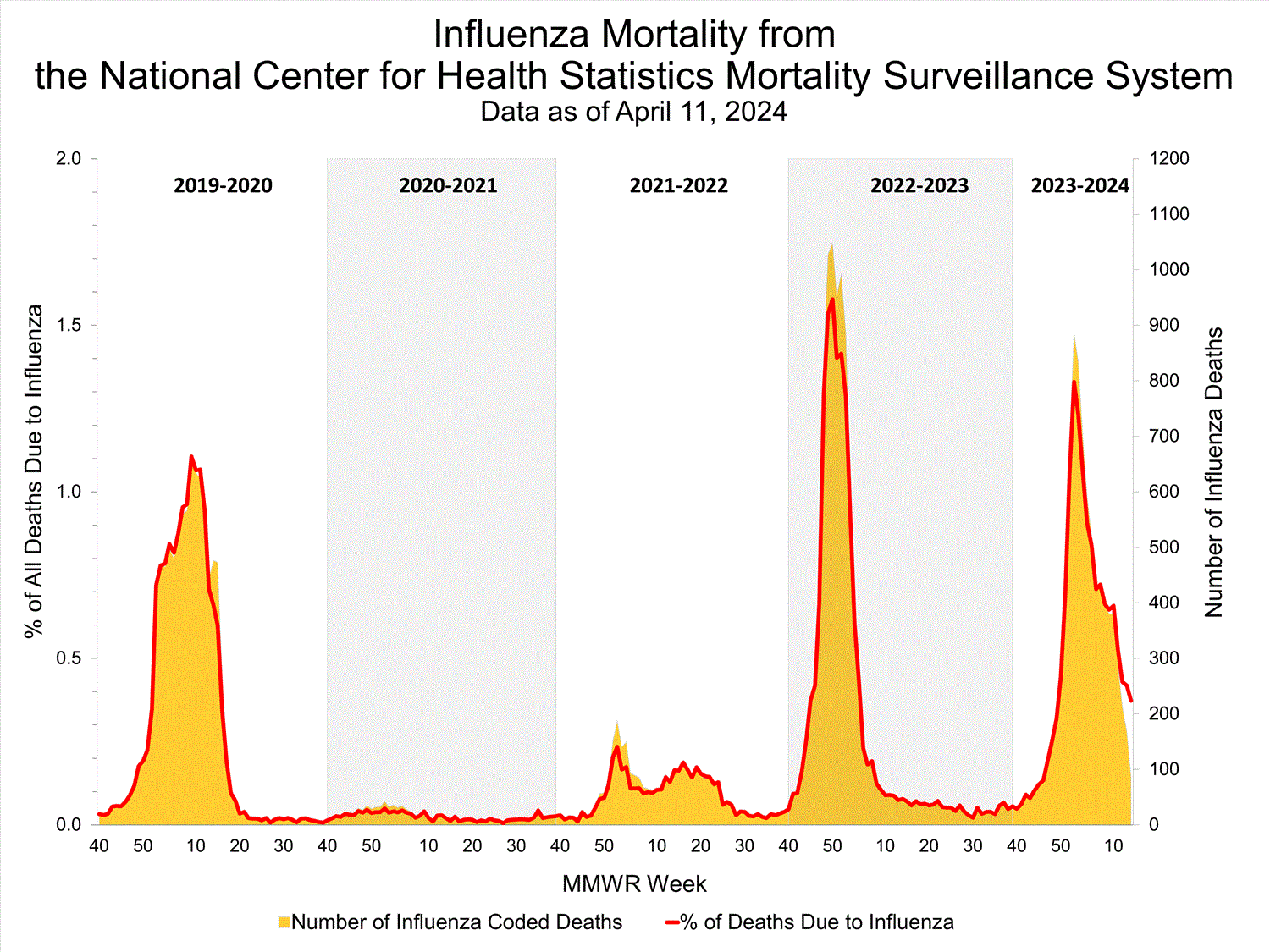
National Center for Health Statistics (NCHS) Mortality Surveillance
Based on NCHS mortality surveillance data available on April 11, 2024, 0.4% of the deaths that occurred during the week ending April 6, 2024 (Week 14), were due to influenza. This percentage remained stable (< 0.1 percentage point change) compared to Week 13. The data presented are preliminary and may change as more data are received and processed.
Additional pneumonia, influenza and COVID-19 mortality surveillance information for current and past seasons:
Surveillance Methods | FluView Interactive
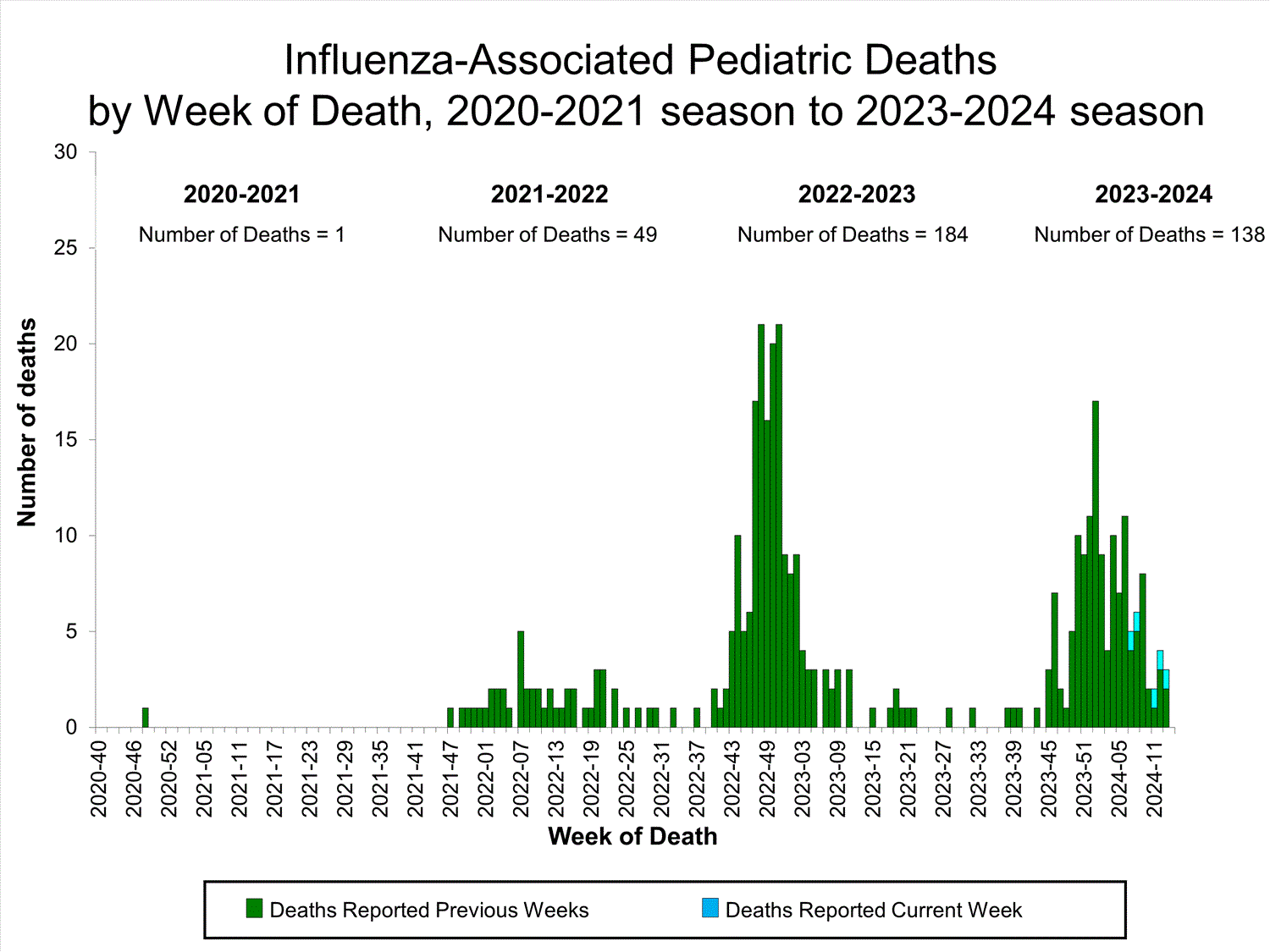
Influenza-Associated Pediatric Mortality
Five influenza-associated pediatric deaths occurring during the 2023-2024 season were reported to CDC during Week 14. The deaths occurred between Week 7 (the week ending February 17, 2024) and Week 13 (the week ending March 30, 2024). Two deaths were associated with influenza A viruses for which no subtyping was performed and three deaths were associated with influenza B viruses with no lineage determined.
Additional pediatric mortality surveillance information for current and past seasons:
Surveillance Methods | FluView Interactive
Trend Indicators
Increasing: 
Decreasing: 
Stable: 
Indicators Status by System
Clinical Labs: Up or down arrows indicate a change of greater than or equal to 0.5 percentage points in the percent of specimens positive for influenza compared to the previous week.
Outpatient Respiratory Illness (ILINet): Up or down arrows indicate a change of greater than 0.1 percentage points in the percent of visits due to respiratory illness (ILI) compared to the previous week.
NHSN Hospitalizations: Up or down arrows indicate change of greater than or equal to 5% of the number of patients admitted with laboratory-confirmed influenza compared to the previous week.
NCHS Mortality: Up or down arrows indicate change of greater than 0.1 percentage points of the percent of deaths due to influenza compared to the previous week.
Reference Footnotes
1U.S. Influenza Surveillance: Purpose and Methods (2023 Oct). Centers for Disease Control and Prevention. https://www.cdc.gov/flu/weekly/overview.htm#ILINet.
2Grohskopf LA, Blanton LH, Ferdinands JM, Chung JR, Broder KR, Talbot HK. Prevention and Control of Seasonal Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices — United States, 2023–24 Influenza Season. MMWR Recomm Rep 2023;72(No. RR-2):1–25. DOI: http://dx.doi.org/10.15585/mmwr.rr7202a1
3Influenza Antiviral Medications: Summary for Clinicians (2023 Sept). Centers for Disease Control and Prevention. https://www.cdc.gov/flu/professionals/antivirals/summary-clinicians.htm.
Additional National and International Influenza Surveillance Information
FluView Interactive: FluView includes enhanced web-based interactive applications that can provide dynamic visuals of the influenza data collected and analyzed by CDC. These FluView Interactive applications allow people to create customized, visual interpretations of influenza data, as well as make comparisons across flu seasons, regions, age groups and a variety of other demographics.
National Institute for Occupational Safety and Health: Monthly surveillance data on the prevalence of health-related workplace absenteeism among full-time workers in the United States are available from NIOSH.
U.S. State and local influenza surveillance: Select a jurisdiction below to access the latest local influenza information.
World Health Organization:
Additional influenza surveillance information from participating WHO member nations is available through
FluNet and the Global Epidemiology Reports.
WHO Collaborating Centers for Influenza:
Australia, China, Japan, the United Kingdom, and the United States (CDC in Atlanta, Georgia)
Europe:
The most up-to-date influenza information from Europe is available from WHO/Europe and the European Centre for Disease Prevention and Control.
Public Health Agency of Canada:
The most up-to-date influenza information from Canada is available in Canada’s weekly FluWatch report.
Public Health England:
The most up-to-date influenza information from the United Kingdom is available from Public Health England.
Any links provided to non-Federal organizations are provided solely as a service to our users. These links do not constitute an endorsement of these organizations or their programs by CDC or the Federal Government, and none should be inferred. CDC is not responsible for the content of the individual organization web pages found at these links.
A description of the CDC influenza surveillance system, including methodology and detailed descriptions of each data component is available on the surveillance methods page.