 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: mmwrq@cdc.gov. Type 508 Accommodation and the title of the report in the subject line of e-mail. Update: Perinatally Acquired HIV/AIDS -- United States, 1997Perinatal transmission of human immunodeficiency virus (HIV) accounts for virtually all new HIV infections in children (1). Through 1993, an estimated 15,000 HIV-infected children were born to HIV-positive women in the United States (2). In 1994, clinical trials demonstrated a two-thirds reduction in the risk for perinatal transmission associated with treatment of HIV-infected pregnant women and their infants with zidovudine (ZDV) therapy (3). The Public Health Service (PHS) issued guidelines for the use of ZDV to reduce perinatal transmission in August 1994 and for universal HIV counseling and voluntary testing of pregnant women in July 1995 (3,4). This report describes increases in HIV testing and use of ZDV treatment among HIV-infected mothers and a continued substantial decline in the incidence of acquired immunodeficiency syndrome (AIDS) during 1992-1996 among children who were infected through perinatal HIV transmission (5). * For states that conduct HIV surveillance, characteristics were examined for children born to HIV-infected mothers (i.e., perinatally exposed) during 1993-1996. Children were classified into one of four categories: those with AIDS, those with HIV infection but without AIDS, those who were uninfected, and those of indeterminate infection status. Timely ascertainment of HIV infection status of children born in 1995 for HIV reporting states versus AIDS-only reporting states was assessed in comparison to estimates of the number of births to HIV-infected women from the National HIV Serosurvey of Childbearing Women (SCBW) (2). Trends in perinatally acquired AIDS incidence were analyzed by quarter year of diagnosis from January 1984 through March 1997 and were adjusted for reporting delays with reclassification of cases initially reported with no identified risk (1). Evaluation of efforts to reduce perinatal transmission following issuance of the PHS guidelines was restricted to analysis of the estimated incidence of perinatally acquired AIDS among infants (aged less than 1 year) by year of birth. To control for changing birth rates, rates of perinatally acquired AIDS for infants per 100,000 births were calculated by using natality data from CDC's National Center for Health Statistics for births from 1992 through June 1995 (the latest birth cohort for which estimates are reliable). Perinatal HIV Surveillance A total of 29 states conducted surveillance for HIV infection among children. These states reported 28% of the cumulative perinatally acquired AIDS cases through September 1997. In these states, perinatally exposed children are monitored to determine their HIV-infection and AIDS status, dates of maternal HIV tests, receipt of prenatal care, and maternal and neonatal use of ZDV and other antiretrovirals during pregnancy. Of children born to HIV-infected mothers from 1993 through 1996, these states reported 4325 children who had perinatally acquired AIDS (344) or HIV infection but without AIDS (487) or who were uninfected or indeterminate (3494). Since the PHS guidelines were issued in August 1994, 2027 (87%) HIV-infected mothers of children reported in these states had HIV infection diagnosed before or at the child's birth and 179 (8%) after the child's birth; the timing of HIV infection diagnosis was unknown for 121 (5%) (Figure_1). For children born from 1994 through 1996, the proportion of HIV-infected mothers who were prescribed prenatal ZDV increased from 24% to 64% (Figure_2). Compared with the expected number of children born to HIV-infected women estimated from the SCBW, as of September 1997, states with HIV surveillance ** identified a median of 64% of children born in 1995 compared with a median of 3% in states with AIDS reporting only. Characteristics of Perinatally Acquired AIDS Cases As of September 30, 1997, perinatal transmission of HIV accounted for 7310 (1%) of the 626,334 total AIDS cases in adults and children reported to CDC by state and territorial health departments. Perinatally acquired cases have been reported from 48 states, the District of Columbia, Puerto Rico, and the Virgin Islands. Five states and/or territories accounted for 64% of all perinatally acquired AIDS cases: New York (27%); Florida (17%), New Jersey (9%), California (6%), and Puerto Rico (5%). The Northeast (44%) and the South (36%) accounted for most (80%) such cases; 85% of cases were diagnosed in metropolitan areas with a population of greater than 500,000 persons and 9% in metropolitan areas with populations of 50,000-500,000 persons. Of the 7310 children with perinatally acquired AIDS, 4461 (61%) were non-Hispanic black, 1723 (24%) were Hispanic, 1057 (14%) were non-Hispanic white, and 54 (less than 1%) were either Asian/Pacific Islander or American Indian/Alaskan Native; 15 were of unknown race/ethnicity. The median age at diagnosis was 17 months, with 40% of cases diagnosed in children aged less than 1 year; 47%, in children aged 1-5 years; and 13%, in children aged greater than or equal to 6 years. Trends in Perinatally Acquired AIDS From 1984 through 1992, the estimated number of children with perinatally acquired AIDS diagnosed each year increased, then declined 43% during 1992-1996 (Figure_3). From 1992 to 1996, declines were similar by race/ethnicity, regions of the United States, and in urban and rural areas (Table_1). Declines were largest among children for whom AIDS was diagnosed at younger ages (less than 5 years). When the analysis was restricted to children diagnosed with perinatally acquired AIDS at age less than 1 year, for birth years 1992 through 1995, the estimated numbers of cases diagnosed in infants was highest among 1991-1992 birth cohorts, then declined 42% from the first half of the 1992 birth cohort (n=172) to the first half of the 1995 birth cohort (n=100). From the first half of the 1992 birth cohort to the first half of the 1995 birth cohort, the incidence declined 39%, from 8.4 per 100,000 births to 5.1. Reported by: State, territorial, and local health depts. Div of HIV/AIDS Prevention-Surveillance and Epidemiology, National Center for HIV, STD, and TB Prevention, CDC. Editorial NoteEditorial Note: This report documents the rapid implementation of recommended HIV counseling and voluntary testing practices and the increasing use of ZDV therapy by health-care providers and use of these services and care by HIV-infected mothers in the United States. The implementation of these recommendations has been temporally associated with a substantial and geographically widespread decline in perinatally acquired AIDS in the United States among all racial/ethnic groups and in both urban and rural areas, particularly since 1994, most likely reflecting the effectiveness of ZDV in reducing perinatal HIV transmission. The findings from states that conduct HIV surveillance indicate that most HIV-infected mothers were tested for HIV before their child's birth and confirm the effectiveness of current PHS guidelines for routine HIV counseling and voluntary testing of pregnant women. Previous assessments also have demonstrated high acceptance levels following counseling by informed providers (4). Documentation of the increasing use of ZDV therapy among mothers following publication of PHS guidelines is consistent with other assessments noting the increased use of ZDV by pregnant HIV-infected women and their newborns that was associated with reduced rates of perinatal transmission (6). Declines in perinatally acquired AIDS began before 1994, most likely reflecting increased use of ZDV to treat HIV-infected women (6). Other factors possibly contributing to the decline include decreases in the number of HIV-infected women giving birth and increases in use of prophylaxis for Pneumocystis carinii pneumonia (PCP) and in antiretroviral treatment for HIV-infected children. However, from 1992 to 1994, the number of children born to HIV-infected mothers was relatively stable (i.e., 6000-7000 per year) (2). At the same time, incidence of PCP among infants did not decrease substantially (5). Because many new antiretroviral agents have not been approved for use in children, the recent declines in perinatally acquired AIDS probably do not yet reflect potent combination therapy with protease inhibitors (7). Even though this report confirms the effectiveness of prevention efforts, the continued incidence of perinatally acquired AIDS among infants documents ongoing perinatal transmission and underscores the need for strategies to ensure that women receive adequate prenatal care, timely HIV counseling, and voluntary testing; gain access to HIV-related care and services; receive chemoprophylaxis to reduce perinatal transmission; and avoid breastfeeding. These findings especially emphasize the need to focus on increasing access to care and providing prevention services to minority populations, among whom rates of AIDS have been highest (i.e., non-Hispanic blacks and Hispanics) (1). Through the Ryan White CARE Act, the U.S. Congress allocates resources for care and services for HIV-infected persons. This act requires that states evaluate their perinatal HIV-prevention efforts through the use of HIV-infection and AIDS surveillance. The most timely evaluation of prevention efforts in states with and without HIV surveillance is analysis of AIDS incidence among infants. States should evaluate trends in rates of perinatally acquired AIDS among infants per 100,000 births. Data on perinatal AIDS incidence can assist states in the identification of reasons for continued perinatal transmission, including missed opportunities for prevention or failures of recommended therapy (e.g., because of antiviral resistance or inadequate adherence). CDC recommends that, as an extension of AIDS surveillance programs, all states and territories conduct surveillance for perinatal HIV exposure with follow-up to determine HIV-infection and AIDS status. Surveillance for HIV exposure and infection and AIDS would enable timely and complete monitoring of the effectiveness of perinatal prevention efforts (8); HIV incidence trends; identification of groups in which prevention strategies are less successful; evaluation of the impact of ZDV on perinatal HIV incidence; assessment of resources required to provide care for HIV-exposed children; the timeliness of receipt of HIV-related care; and potential short- or long-term adverse effects of in utero exposure to ZDV and other antiretroviral therapy. In addition, the Council of State and Territorial Epidemiologists has recommended that all states conduct surveillance for pediatric HIV/AIDS and perinatal exposure and, in 1995, declared pediatric HIV infection a nationally notifiable disease. The American Academy of Pediatrics also supports surveillance efforts to monitor perinatally exposed and infected children (9). Substantial decreases in perinatal transmission of HIV have been documented in the United States and in some European countries (10); however, most HIV-infected children are born in developing countries. The Joint United Nations Program on HIV/AIDS (UNAIDS) estimates that each year 350,000 children in developing countries are infected with HIV through perinatal transmission. The reduction of perinatal transmission of HIV in the United States underscores the need to identify and evaluate safe, effective regimens that are logistically and economically feasible in developing countries. References
* Single copies of this report will be available until November 21, 1998, from the CDC National AIDS Clearinghouse, P.O. Box 6003, Rockville, MD 20849-6003; telephone (800) 458-5231 or (301) 519-0023. ** Pediatric HIV reporting areas that conducted the SCBW were Alabama, Arizona, Arkansas, Colorado, Connecticut, Indiana, Louisiana, Michigan, Minnesota, Mississippi, Missouri, Nevada, New Jersey, North Carolina, Ohio, Oklahoma, Oregon, South Carolina, Tennessee, Texas, Utah, Virginia, West Virginia, Wisconsin, and Wyoming. The four HIV reporting areas that did not conduct the SCBW were Idaho, Nebraska, North Dakota, and South Dakota. Figure_1 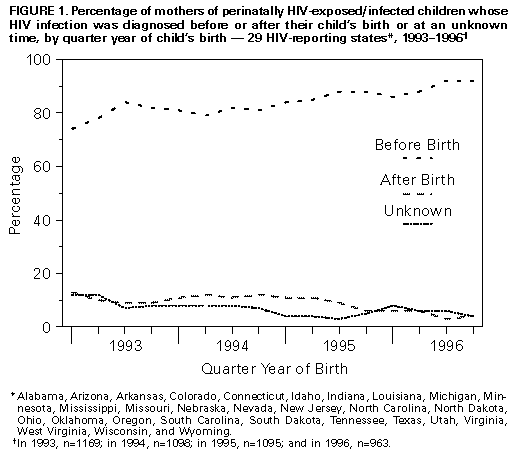 Return to top. Figure_2 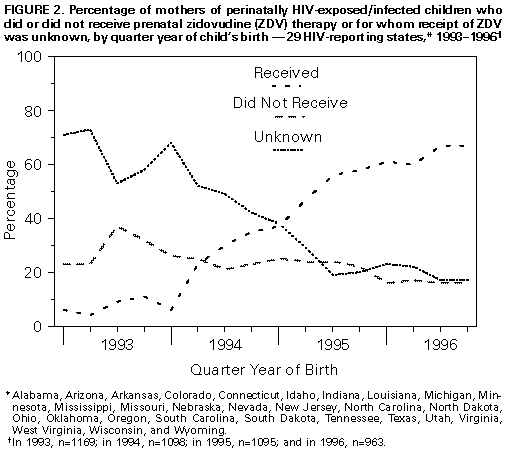 Return to top. Figure_3 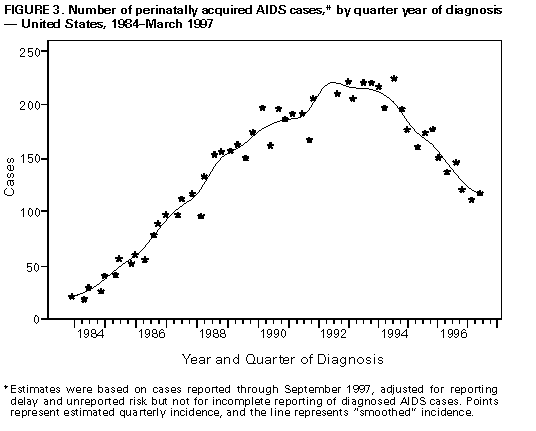 Return to top. Table_1 Note: To print large tables and graphs users may have to change their printer settings to landscape and use a small font size.
TABLE 1. Estimated number of children with perinatally acquired AIDS, by selected
characteristics, year of diagnosis, and percentage change from 1992 to 1996 -- United States,
1992-1996 *
================================================================================================
Year
-----------------------------------
Characteristic 1992 1993 1994 1995 1996 % Change 1992 to 1996
------------------------------------------------------------------------------------------------
Race/Ethnicity +
White,non-Hispanic 133 126 92 95 67 -50%
Black,non-Hispanic 566 531 522 415 331 -42%
Hispanic 195 195 166 146 111 -43%
Age at AIDS diagnosis
<5 years 733 693 613 459 360 -51%
>=5 years 168 169 179 202 156 - 7%
Region&
Northeast 361 379 315 265 212 -41%
South 362 315 332 243 223 -38%
Midwest 60 74 54 67 30 -50%
West 67 58 65 60 35 -48%
Metropolitan statistical area
500,000 population 748 732 675 558 450 -40%
50,000-500,000 population 102 75 75 62 41 -60%
<50,000 population 51 53 42 39 22 -57%
------------------------------------------------------------------------------------------------
* Diagnosed through 1996 and reported through September 1997 adjusting for reporting delays and
unreported risk.
+ Numbers for other racial/ethnic groups were too small for meaningful analysis.
& Northeast=Connecticut, Maine, Massachusetts, New Hampshire, New Jersey, New York,
Penn-sylvania, Rhode Island, and Vermont; South=Alabama, Arkansas, Delaware, District of
Columbia, Florida, Georgia, Kentucky, Louisiana, Maryland, Mississippi, North Carolina,
Oklahoma, South Carolina, Tennessee, Texas, Virginia, and West Virginia; West=Alaska,
Arizona, California, Colorado, Hawaii, Idaho, Montana, Nevada, New Mexico, Oregon, Utah,
Washington, and Wyoming; and Midwest=Illinois, Indiana, Iowa, Kansas, Michigan, Minnesota,
Missouri, Ne-braska, North Dakota, Ohio, South Dakota, and Wisconsin.
================================================================================================
Return to top. Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to mmwrq@cdc.gov.Page converted: 09/19/98 |
|||||||||
This page last reviewed 5/2/01
|